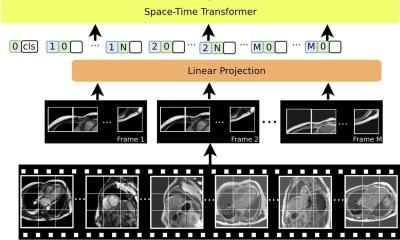
News • Sticker health
Ultrasound-based pacemaker to noninvasively steady the heart
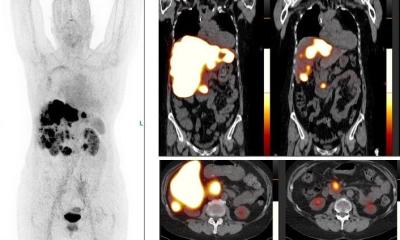
Engineers have developed a noninvasive pacemaker that stimulates the heart using ultrasound. The design could one day provide a surgery-free alternative to traditional cardiac implants.