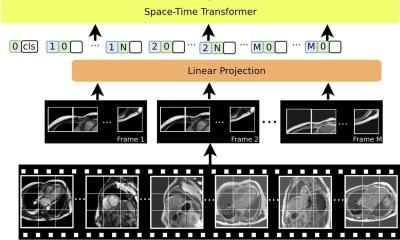
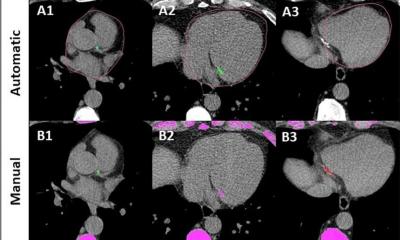
Article • Advancing kidney disease investigation
Digital spatial profiling: new ways for diagnostic histopathology
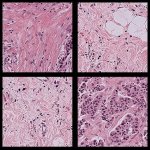
Digital spatial profiling (DSP) is emerging as a powerful technology in helping specialists investigate complex kidney disease, according to a leading expert. Professor Renate Kain believes spatial profiling adds significantly to systems biology approaches that will transform diagnostic histopathology. However, she warns that the adoption and effective utilization of the technique is critically…