News • Impact beyond weight loss
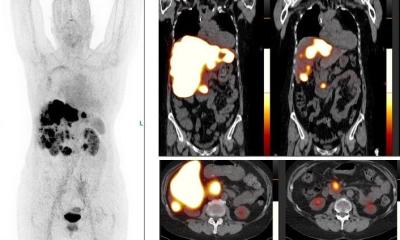
Whole-body PET/CT reveals metabolic changes after bariatric surgery
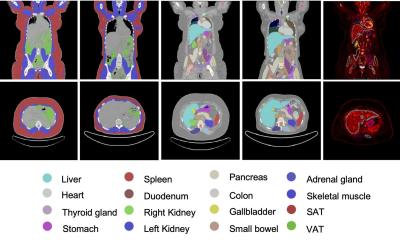
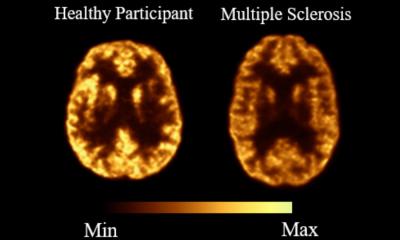
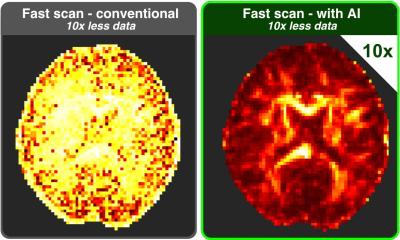
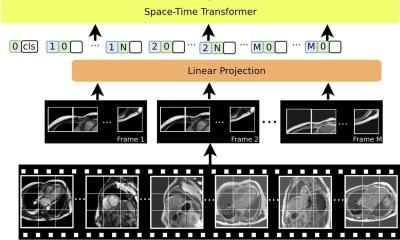
A novel PET imaging framework reveals that bariatric surgery impacts not only body weight, but also the metabolic function of organs. These insights could support more precise treatment monitoring.