News • Artificial urinary sphincter
Trial puts new incontinence implant to the test
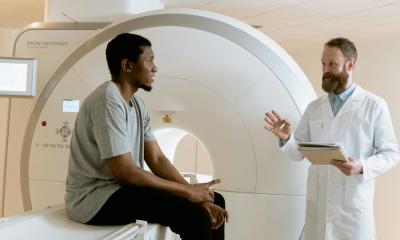
Urologists at Vanderbilt Health are enrolling men with stress urinary incontinence (SUI) in a clinical trial named SOPHIA2 to assess the safety and efficacy of the UroActive implant, following an investigational device exemption clearance from the U.S. Food and Drug Administration and the French National Agency for the Safety of Medicines and Health Products.

Image credit: Vanderbilt Health
SUI, or involuntary urinary leakage, affects an estimated 40 million Americans and 90 million Europeans. SUI significantly impacts quality of life, as it can be debilitating, and often leads to depression, low self-esteem and social stigma.
The implant is placed around the urethral duct and is controlled based on the patient's activity, without the need for complex manipulation, intending to provide patients with ease of use and a better quality of life than current options.
“We have seen firsthand the shortcomings of current SUI treatment options for our male and female patients,” said study co-principal investigator Melissa Kaufman, MD, PhD, the Patricia and Rodes Hart Professor of Urologic Surgery and chief of Reconstructive Urology and Pelvic Health at Vanderbilt Health. “That’s why we’re so excited to be leading the SOPHIA2 trial, as it’s showing promise to provide significant improvements in addressing these issues.”
Kaufman is leading the trial in the U.S. along with Andrew Peterson, MD, professor of Urology at Duke University School of Medicine.
The trial will enroll approximately 140 male patients with SUI across clinical trial sites in the U.S. and France. To be eligible for enrollment, patients must be at least 22 years old, have a primary diagnosis of SUI for at least 6 months, and have been deemed an appropriate candidate for surgery.
The primary end point is the responder rate at 26 weeks following device activation. The study’s secondary end point is the proportion of patients who experience a change in overall impression of improvement from baseline to 26 weeks.
Data from the first-in-human SOPHIA study of the UroActive implant in men was presented at the American Urological Association 2024 Annual Meeting in San Antonio, showing positive outcomes on all primary and secondary end points.
According to the investigators, the devices were successfully implanted and activated in all six patients, and there were no explants nor revisions required. There was one severe adverse event, consisting of a hematoma after surgery with slow urinary stream, which was self-resolved.
The UroActive device is also under investigation in women with SUI and has demonstrated comparable results in the clinical feasibility study.
Source: Vanderbilt University Medical Center
13.03.2026