News • Critical role of NFAT
Preventing pregnancy complications with new immune insights
Discovery sheds light on preeclampsia, implantation failure and inadequate blood supply to the placenta

Image credit: Jennifer Alsabrook-Turner/UAB
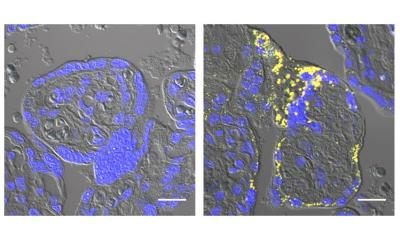
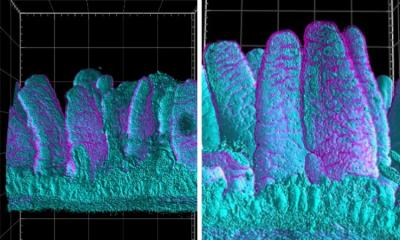
Early pregnancy depends on a remarkable act of coordination. Before the placenta can nourish a growing fetus, the embryo must securely “land” and connect with the mother’s blood supply — a process guided by a specialized group of immune cells called uterine natural killer cells, or uNK cells.
A new peer-reviewed study from the University of Alabama at Birmingham, supported by the National Institutes of Health and published in Science Translational Medicine, has uncovered a critical role in successful pregnancies for an immune switch called NFAT that helps uNK cells take up residence in the uterus and perform that critical guidance work. When the switch is turned down, fewer natural killer cells make their way to the uterus — and pregnancy complications may follow.
“This discovery opens an entirely new window into how the immune system supports healthy pregnancy,” said Paige Porrett, M.D., Ph.D., lead author of the study and professor of surgery and obstetrics and gynecology, as well as the Vera Hauptfeld‑Dolejsek Endowed Professor of Transplant Immunology in UAB’s Heersink School of Medicine. “Seeing NFAT act as a residency switch in uterine NK cells was a surprise, and it gives us a concrete lead on why some pregnancies veer toward complications. This opens up a whole new world of research.”
The findings offer fresh insight into the biology behind preeclampsia, implantation failure, inadequate placental blood flow and some early pregnancy losses — conditions that affect many pregnant patients, not only those who have undergone organ transplantation. The team’s expertise in uterus transplantation provided a unique opportunity to study the immune environment in pregnancy. But the mechanism they uncovered appears to be a fundamental part of human placental development. “This isn’t just a transplant story,” Porrett said. “We used transplant as a lens, but the biology may map onto normal pregnancies and high‑risk ones. The benefits of this work are potentially for all pregnant patients.”
UAB scientists discovered that NFAT controls whether uNK cells become “tissue‑resident,” a necessary step that allows them to remain in the uterine lining and remodel maternal blood vessels for early placental growth. Until now, NFAT was known for its role in T cells but had never been described in the context of uterine NK cells or pregnancy. “This is a new pathway for immune cells to become resident that we didn’t know about before,” Porrett said. “We now show in humans that the biology we’ve only ever seen in mice applies. As a result, this opens a whole new world of research.”
We need to validate causality and understand how other uterine cell types respond. That’s the hard work ahead — and it’s how we turn a discovery into better outcomes
Paige Porrett
The team used single‑cell RNA sequencing, a powerful modern tool that measures the activity of every gene in individual cells. This level of detail allowed researchers to see gene programs shift across thousands of cells — something older approaches could not capture, especially in tissues as dynamic as the uterus. “Single‑cell sequencing let us see, cell by cell, the gene programs that were changing,” Porrett said. “You need that granularity to identify a pathway like this.”
The study also raises important scientific questions about tacrolimus, a standard immunosuppressive medication taken by nearly all solid‑organ transplant recipients. While tacrolimus is highly effective at preventing rejection — and prior studies have long shown it does not increase birth‑defect risk — the new findings suggest that lowering the NFAT signal using this medication may raise the risk of placental complications. “Tacrolimus works incredibly well, but now we’ve identified a biological reason to reconsider how we use it during pregnancy,” Porrett said. “We have to do the hard work of finding safer alternatives for pregnant patients while still preventing rejection.”
Researchers emphasize that no medication changes should be made based on this study alone. Instead, the findings provide a mechanistic foundation for the next phase of research: identifying alternative drugs or timing strategies that protect both the pregnancy and the transplanted organ. “It’s a balance,” Porrett said. “Tacrolimus is a workhorse, and any change must be evidence‑based.”
Beyond transplant recipients, the researchers say the work opens new avenues for understanding pregnancy complications in patients who are not immunocompromised. The team plans to map how immunosuppressive drugs affect other immune and non‑immune cell types in the uterus — including stromal and epithelial cells — to understand the broader biological landscape. “These human data are powerful, but they’re largely associative,” Porrett said. “We need to validate causality and understand how other uterine cell types respond. That’s the hard work ahead — and it’s how we turn a discovery into better outcomes.”

Image credit: Jennifer Alsabrook-Turner/UAB
Porrett’s co-authors for the study, “Inhibition of NFAT after human uterus transplant promotes loss of tissue resident NK cells and attendant pregnancy complications,” include from UAB Rebecca Asiimwe, Brittney Knott, Morgan E. Greene, Emma D. Wright, Markayla Bell, Daniel Epstein, Stefani D. Yates, Samantha Fry, Emily Boydston Procópio, Stephanie Clevenger, Jayme E. Locke, Brian E. Brocato, Constantine M. Burgan, Richard Burney, Nitin Arora, Virginia E. Duncan, Holly E. Richter, Deidre Gunn and Shawn C. Little; Michael V. Gonzalez, University of Pennsylvania; and Aharon G. Freud, The Ohio State University.
Support came from the National Institutes of Health grants R01AI177369, R01AI145905 and R01CA208353; UAB Center for Women’s Reproductive Health; UAB AMC21 grants; startup funding from the UAB Heersink School of Medicine; and American Cancer Society Research Scholar Grants #RSG-23-1153857-01-IBCD, F31HD114429 and T32GM135028.
Source: University of Alabama at Birmingham
12.03.2026