Article • High-sensitivity troponin I assay
Speeding up diagnosis of myocardial infarction
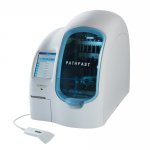
The new generation troponin I assay unveiled by Beckman Coulter is helping improve heart attack diagnosis. Delivering high sensitivity, and rapid result turnaround times, every element of the Access hsTnI assay has been redeveloped and updated, including the antibodies and the paramagnetic particles used. As specialists in developing and manufacturing products that simplify and automate complex…