Article • Advances in cancer risk assessment and prevention
French oncologists pick up the pace after pandemic time-out
The pandemic-imposed pause for oncology care is coming to an end: Real-life data and personalised screening techniques are set to improve cancer prevention and patients’ quality of life. In a dedicated press conference in Paris, French oncologists presented promising research that might bring hope for many cancer patients.
Report: Mélisande Rouger

Image source: Unicancer
French cancer centres are catching up after the coronavirus pandemic put oncology interventions on hold in 2020, and the spirit of a fresh start was prevalent at an event organised by Unicancer, a federation regrouping 19 leading institutions specialising in oncology. ‘We’re getting out of it, after experiencing delays that have been detrimental to patients’ recovery,’ Unicancer President Jean-Yves Blay, a professor of medical oncology at Claude Bernard University in Lyon, said.
After all, the pandemic left a considerable dent in cancer care: French facilities registered a 7% diagnostic delay for oncology patients in the aftermath of the Covid-19 outbreak in 2020. One year later, Unicancer centres, which treat about 20% of patients with cancer in France, reported a 6% increase in activity.
Making the most of the available data
Unicancer presented a series of proposals to better fight cancer, which remains the leading cause of death in France, with nearly 400,000 new cases and more than 157,000 deaths each year. Proposals target healthcare professions’ attractiveness and remuneration and offer strategies on how to boost follow-up and prevention. The experts agreed that 40% of cancers could be avoided if current knowledge about risk factors was translated into effective public health strategies. ‘We want to develop highly connected research points to improve prevention from risk factor identification to screening,’ Blay said. ‘Treatment follow-up of cancers in children and rare cancers, which remain difficult to treat, is also one of our priorities.’
Image source: Adobe Stock/peterschreiber.media
Having this huge amount of real-life health data will help us better predict or evaluate treatment
Jean-Yves Blay
With over 100 studies enrolling 8,000 patients in 2021, Unicancer touches on several aspects of research beyond the clinical spectrum, focusing on issues such as patients’ quality of life and how to make use of data which is already available.
A few months ago, the organisation launched a pilot project with Health Data Hub, a platform dedicated to facilitating the sharing of health data from a wide variety of sources, to help accelerate research. The program, called UNIBASE, aims to harness data from electronic patient records and create a collection of cancer reference databases within three years. ‘Having this huge amount of real-life health data will help us better predict or evaluate treatment,’ Blay said.
Personalising screening and patient involvement
Underlining the historical weakness of France’s public health culture, the latest report of the French National Audit Office on prevention policies mentioned ‘mediocre results’, despite significant financial efforts similar to those of European neighbours.1 For example, screening remains problematic in France. National programmes are organised for breast cancer, colorectal cancer and cervical cancer, but participation remains insufficient.

Image source: EURORDIS
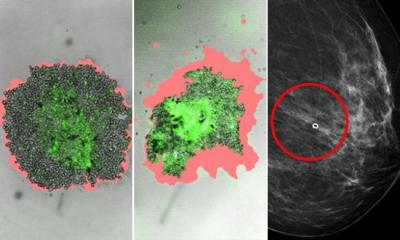
To improve screening efforts, researchers are looking to more personalised approaches, for example in breast cancer screening programmes. To this end, Unicancer is taking part in MyPeBS, an EU-funded randomised controlled trial that aims to assess the value of personalised breast cancer screening for women aged 40 to 70 in Europe. The study plans to determine whether risk-based cancer screening in these women is non-inferior to the standard screening programme currently offered in Belgium, France, Israel, Italy, and the United Kingdom. The project will follow 85,000 women over four years and could mean a great advance, Blay explained. ‘This study is a revolution in screening management. If completed, it will transform the routine,’ he said.
Patient empowerment is another promise of personalised cancer medicine. The aim is to involve the patient in clinical decision-making, another speaker highlighted in the conference. ‘We’re witnessing a cultural change and a reconstruction of clinical decision-making,’ Ariane Weinman, a representative at EURORDIS, a non-governmental alliance of rare disease patient organisations, said. ‘Patients and their families are increasingly involved and informed, sometimes even receive dedicated training to prepare for the experience.’
Patient organisations also increasingly take part in clinical trials, which benefits everyone, according to Weinman. ‘The more involved the patients are, the better they will be able to follow their treatment and support research near the authorities,’ she concluded.
1 https://www.vie-publique.fr/sites/default/files/rapport/pdf/282706.pdf
05.05.2022