Article • New therapies, new questions
To understand an image is to understand the treatment
Conventional chemotherapy in oncology is increasingly yielding to new procedures such as targeted therapy and immunotherapy. This results in changes for radiologists because new procedures require familiarisation with certain imaging to ensure that the treatment process is interpreted correctly. During a recent CT symposium Professor Hans-Christoph Becker, radiologist at Stanford University Medical Centre in California, USA, discussed aspects of such changes.
Report: Wolfgang Behrends

‘To begin with it’s important to understand how the new therapies actually work,’ radiologist Professor Hans-Christoph Becker advised, speaking at the CT-Symposium 2018 held in Garmisch, Germany. Targeted treatment and immunotherapy attack tumours in a very different way from chemotherapy. ‘The point of chemotherapy is to destroy tumour cells. Targeted, i.e. specific cancer therapy works, for example, with specific messenger substances that activate signal paths, and explicitly interfere with the metabolism of tumour cells.’ cells.’ The advantage of the new treatments lies in their higher precision. ‘Chemotherapy is comparatively non-selective,’ Becker explains. ‘It damages tumour cells as well as healthy tissue, which means it causes collateral damage.’ By comparison, targeted therapies attack the tumour cells, which can be identified with high specificity with the help of surface antigens, for instance. This can help to reduce the severity of side effects for patients.
Therefore, the objective for researchers is to make the treatment as precise as possible. ‘Each patient has tumours with a certain, individual combination of characteristics,’ Becker points out. These markers are determined histologically via biopsies. ‘We can now differentiate with much more precision which treatments tumours are most likely to respond to and can therefore target therapy much more individually.’ Treatment success is monitored clinically as well as through imaging.
Size does matter – but so do other aspects
It makes the tumour appear larger on the image. What it really means though is that the treatment is working
Hans-Christoph Becker
The high precision of therapies, however, also entails some new difficulties: ‘With chemotherapy, treatment success is usually confirmed by the fact that a tumour shrinks,’ the expert adds. ‘This is different with the new targeted therapies. The objective is not necessarily only the destruction of the tumour tissue but also a significant impact on its metabolism.’ This reduces the blood supply to the tumour. ‘It leads to effects which look different from those of conventional chemotherapy.’ Tumours are usually heterogeneous, i.e. they consist of different cell lines. Targeted therapy has become so specific that sometimes only some of these lines respond to it, but not others. ‘This leads to a peculiar response where some tumours and metastases may shrink, whilst others even grow at the same time.’
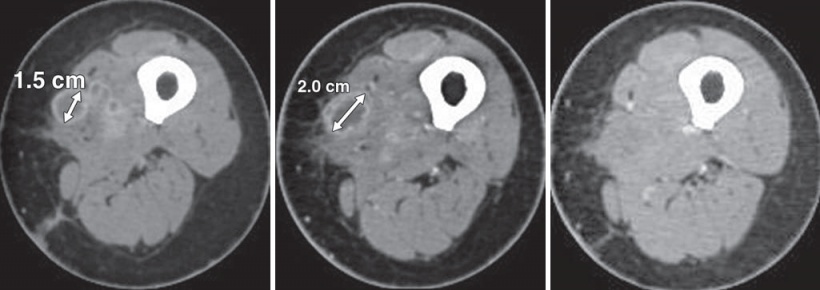
With other procedures, such as immunotherapy, the tumour initially clearly looks bigger in the CT image – however, this does not mean that the tumour is not responding to treatment. ‘To the contrary, this is a classic phenomenon of immunotherapy and the result of a deliberately induced inflammatory reaction,’ Becker explains. ‘Some of these therapies release the handbrake of the body’s immune cells. The immune system then attacks the tumour with leucocytes, which makes the tumour appear larger on the image. What it really means though is that the treatment is working.
‘Radiologists must be able to recognise these effects to evaluate the CT images correctly.’ Understanding how different therapies work is also essential for correct interpretation of images. ‘The interaction between pathology, tumour treatment and image analysis is increasingly complex,’ says Becker. ‘Linking these areas in a meaningful way will be among the big challenges in the future.’
AI analysis presents great potential
Automated evaluation of such imaging phenomena is also conceivable. Artificial intelligence (AI) could detect the specific reactions of tumours in the data sets and therefore measure treatment success. Stanford University Medical Center is already working on the development of such algorithms. ‘The procedure is still in its infancy, but it’s very exciting. It is likely that AI-supported evaluation will soon have a major impact in radiology.’
The ‘new’ cancer therapies referred to here are actually not that new: ‘Hormone therapy was already in clinical use 20 years ago,’ Becker points out. ‘However, subsequently, more and more therapies have been discovered and the number of licensed targeted therapies also continues to increase. ‘Many different drugs are currently being trialled, and studies into combining these with conventional treatment, as well as doseranging studies will continue to run.’ Fundamentally, the new therapies can be used anywhere that conventional chemotherapy is used.
Profile:
From 2001-2014 Professor Hans-Christoph Becker was a senior consultant at the Institute for Clinical Radiology at Ludwig-Maximilian-University (LMU), in Munich, Germany. In 2009 he was appointed Professor of Radiology, focusing on non-invasive cancer imaging. About three years ago he received a generous budget to set up a cancer research laboratory at Stanford University, California, USA, similar to the one he established at LMU in Munich.
27.02.2018