Image source: Lòpez-Mateu C, Gómez-Mahiques M, Gil-Terrón FJ et al., Medical Physics 2026 (CC BY-NC-ND 4.0)
News • Biomechanical mapping of tumor growth
New biomarker helps assess glioblastoma aggressiveness
Developed by the ITACA BDSLab, the new biomarker can identify different patterns of tumour growth and independently predict patient survival
Researchers from the Biomedical Data Science Laboratory (BDSLab) at the ITACA Institute of the Universitat Politècnica de València have developed a new method based on magnetic resonance imaging that enables objective quantification of the growth of the most aggressive brain tumours, particularly glioblastoma.
The study, published in the scientific journal Medical Physics, addresses one of the main clinical challenges in the diagnosis and treatment of this tumour: its high capacity to infiltrate healthy brain tissue.
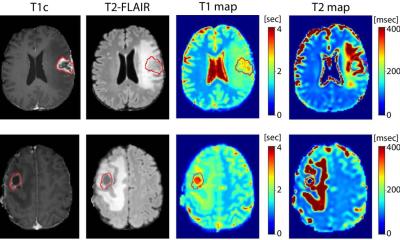
Until now, assessment methods have mainly been based on measuring the increase in tumour size or the displacement of brain structures, without adequately capturing how the tumour grows or its biomechanical impact on the surrounding brain
Carles López Mateu
In their work, the UPV's BDSLab team presents a new biomarker, the Dynamic Infiltration Rate (DIR), capable of identifying different patterns of tumour growth and independently predicting patient survival. "Until now, assessment methods have mainly been based on measuring the increase in tumour size or the displacement of brain structures, without adequately capturing how the tumour grows or its biomechanical impact on the surrounding brain," explains Carles López Mateu, lead author of the study.
The research was carried out by Carles López Mateu, María Gómez Mahiques, F. Javier Gil Terrón, Víctor Montosa i Micó, Juan M. García-Gómez and Elies Fuster García, in collaboration with researchers from Oslo University Hospital.
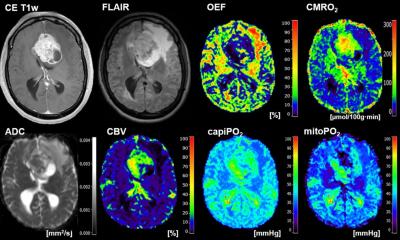
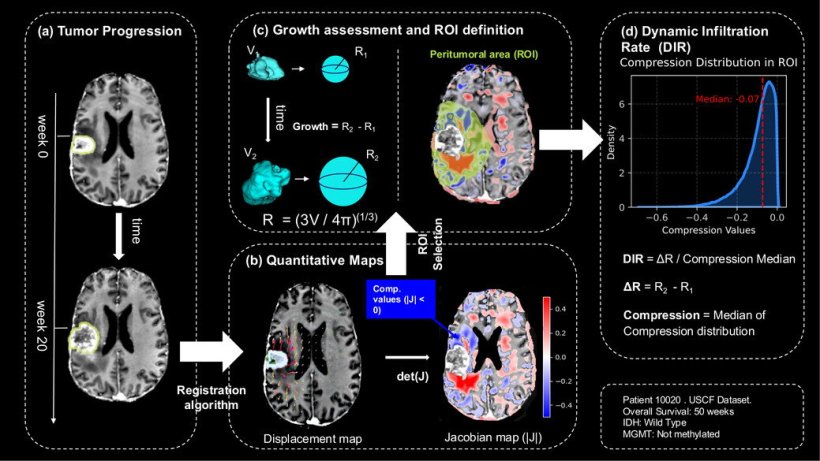
The biomarker developed by the BDSLab-ITACA team combines the tumour's volumetric growth over time with the mechanical effects this growth exerts on adjacent brain tissue. From a longitudinal analysis of magnetic resonance imaging, the researchers have generated tissue compression maps that allow them to assess how the tumour compresses or infiltrates healthy tissue.
The DIR biomarker integrates both phenomena and allows differentiation between more proliferative tumours that compress the brain and more infiltrative tumours that expand without significant compression. "This index allows us to characterise the biological behaviour of the tumour beyond its size and provides key information on its aggressiveness," notes Carles López.

Image source: Universitat Politècnica de València
The method has been validated using both synthetic data and two international clinical cohorts of glioblastoma patients. The results show that the DIR allows for robust stratification of patients according to their prognosis. "Patients with low DIR values have an average survival of 35.2 weeks, compared to 16.0 weeks for those with high values," highlights María Gómez Mahiques, a researcher at ITACA and co-author of the study. These results demonstrate the potential of the DIR as a tool to support clinical decision-making by facilitating a more precise characterisation of tumour aggressiveness.
The study conducted by the UPV team and Oslo University Hospital paves the way for more personalised medicine by enabling therapeutic strategies and follow-up protocols to be tailored to the growth pattern of each tumour. "This is a quantitative, reproducible and non-invasive biomarker, based exclusively on medical imaging, which reinforces the role of biomedical engineering and data science in precision oncology and, at the same time, utilises accessible methodologies that facilitate its future transfer to clinical practice," the authors conclude.
Source: Universitat Politècnica de València
07.04.2026