Article • Immuno-oncological biomarkers
Seeking to augment the value of tumour infiltrating lymphocytes
Measuring tumour infiltrating lymphocytes (TILs) is gaining importance in immunotherapy, but other variables must also be considered to boost prognosis and prediction accuracy, a leading pathologist argued at EBCC 11 last March in Barcelona.
Report: Mélisande Rouger

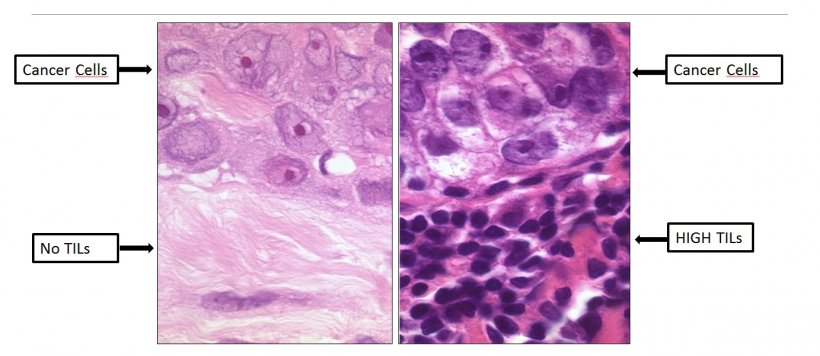
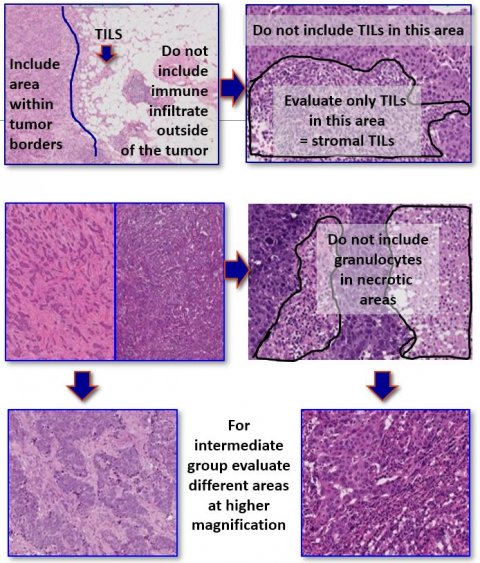
When it comes to prognosis and prediction for immunotherapy, a potentially new variable is emerging – TILs – white blood cells that have left the blood stream to migrate towards tumours and that have always been present in the tissue from where the cancer originates. These can be assessed on haematoxylin and eosin-stained (H&E) slides, and therefore are a marker that can easily be assessed by pathologists worldwide, since only a microscope and one H&E-slide need to be used, Belgian pathologist Roberto Salgado pointed out.
Recommended article
News • Infiltrating tumors
Immune cells predict therapy response in breast cancer
When immune cells invade the tumor, this is usually considered a good sign because the body's own immune system appears to be responding to the cancer. In the case of certain types of breast cancer immune cells, namely so-called tumor-infiltrating lymphocytes (TILs), these can determine survival rates and predict the usefulness of chemotherapy. This was shown by the largest meta study on TIL…
Creating a ‘chemotherapy response map’
‘In general, TILs and immune signatures in breast cancer are strongly correlated,’ he explained. ‘The question is: How can we define added clinical value for TILs with H&E and do we need additional variables, such as immune signatures – and, if yes, how should we use these?’ Carsten Denkert, a leading pathologist from the Charité Hospital in Berlin, combined the number of TILs and PD1 mRNA signatures in what he described as a ‘chemotherapy response map’. In this context, quantity clearly matters, Salgado pointed out. ‘If you have a lot of TILs and PD1 mRNA signatures, your results are excellent. But there is this middle category of TILs with intermediate scores, which is a bit blurry and difficult for pathologists to assess. The question remains how, and whether we should combine these two variables in order to define predictive accuracy of treatments.’
If you have metastatic breast cancer and a lot of TILs, you do excellent in terms of response
Roberto Salgado
The idea of using TILs as a biomarker is gaining weight among pathologists, and evidence on clinical utility is emerging. The moment has come to use TILs in clinical trials and assess even combinations of TILs with immune-gene expression signatures in future clinical trials design, Salgado argued. In a recent study associating TILs with anti-PD(L)-1 response, Salgado and co-researchers demonstrated that a high number of TILs in the metastatic setting means treatment is working, independently from all other variables. ‘If you have metastatic BC and a lot of TILs, you do excellent in terms of response,’ he explained. These results were confirmed in another study on TILs in a nivolumab mTNBC-trial from the Netherlands Cancer Institute (NCI).
The combination of variables seems to be key to enhance accuracy, since patients who have extremely high number of TILs may not all respond to treatment and we need to be able to identify these. Salgado suggests using, for instance, TILs in alliance with genetic profiles to identify these patients. Neoadjuvant setting is quite an opportune moment to predict which patients will present pCR (pathological Complete Remission, i.e. the complete disappearance of cancer after treatment) after chemo + anti-PD1, evidence shows. ‘We do know that if you just give chemotherapy and perform a biopsy after treatment, most of TILs go up. This is also the case in neoadjuvant setting,’ he said. ‘We will have more information available at next major cancer meetings.’
An important question that clinicians must also answer is how to use this information. They should probably consider adding RCB – Residual Cancer Burden, i.e. a measurement of the amount of cancer left after treatment – as an endpoint including the number of TILs, Salgado believes. ‘Can we identify either preoperatively this subcategory or – and this is emerging – can we develop so-called post neoadjuvant trials, in which we can stratify this patient population into treatments on high TILs and RCB with an immune checkpoint and without, depending on the amount of cancer left after treatment?’
Professor Michail Ignitiadis, from the Jules Bordet Institute, recently conducted an enlightening trial in patients receiving trastuzumab/pertuzumab based chemotherapy in the neoadjuvant setting (TRYPHAENA sub study). The results revealed that having a high number of TILs at baseline is a good indication that treatment is working, whereas having a low amount of TILs translates into a worse prognosis. ‘This shows we need this combination of markers, of both a measure of disease after treatment and TILs in this case,’ Salgado pointed out.
The combination of TILs with gene expression profiles can outline three types of categories of response, all triggering different questions. ‘Having a lot of TILs and gene expression profiles is good news; yet can we do even better in terms of enhancing immune reaction? And, if you have a low number of TILs and low expression profiles, how can we enhance the immune system? As for having a lot of TILs and intermediate gene expression, or vice versa, we don’t know what to do with this category. However, these are the majority of our patients,’ Salgado said.
Eventually, TILs must be combined with something else to improve prognosis. In a recent study carried out in a pooled analysis of phase-3 clinical trial in patients with TNBC (Triple Negative Breast Cancer), Salgado and co-researchers have combined TILs and nodal status by tumour size and age. ‘We could accurately define five years of survival of these patients. But what do we do with this information? This is challenging, arguing that this information can be used to develop clinical trials testing, for example deescalating treatment options,’ he said.
Prediction, defined as predicting response to a particular treatment may require multiple sources of information. ‘When you have a high number of TILs, you have good response, but,’ he concluded. ‘you will obtain higher accuracy combining TILs with something else, for instance LDH concentration.’
Profile:
Roberto Salgado is an anatomic pathologist at Department of Pathology in GZA/Antwerp, Belgium. He is also a scientific collaborator at the Breast Cancer Translational Research Laboratory in the Jules Bordet Institute, the Immuno-Task Force of the Breast International Group and the Translational Breast Cancer Genomic and Therapeutics Laboratory of the Peter Mac Callum Cancer Centre in Melbourne, Australia. He chairs the International Immuno-Oncology Biomarkers Working Group, an international consortium of pathologists who develop guidelines and guidance on the assessment of immuno-oncological biomarkers in cancer. Salgado works in close collaboration with the EORTC and is an auditor on molecular pathology/genetic laboratories for the federal Belgian government.
28.12.2018