News • Brain cancer research
New approach could stop glioblastoma growth
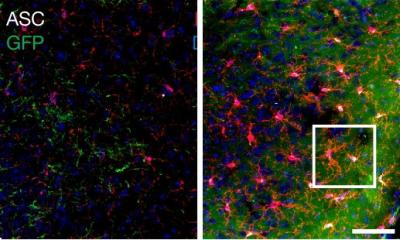
Inhibiting a key enzyme that controls a large network of proteins important in cell division and growth, paves the way for a new class of drugs that could stop glioblastoma, a deadly brain cancer, from growing.
Researchers at Princess Margaret Cancer Centre, the Hospital for Sick Children (SickKids) and University of Toronto (U of T), showed that chemically inhibiting the enzyme PRMT5 can suppress the growth of glioblastoma cells. The inhibition of PRMT5 led to cell senescence, similar to what happens to cells during aging when cells lose the ability to divide and grow. Cellular senescence can also be a powerful tumour suppression mechanism, stopping the unrelenting division of cancer cells.

Credit: The Princess Margaret Cancer Foundation
The results of the study are published in Nature Communications, by lead author, former Postdoctoral Fellow Dr. Patty Sachamitr, SickKids and U of T, and senior authors, Drs. Panagiotis Prinos, Senior Research Associate, Structural Genomics Consortium, U of T, Senior Scientist Cheryl Arrowsmith, the Princess Margaret, and Senior Scientist and Neurosurgeon Peter Dirks, SickKids. The researchers are part of the Stand Up To Cancer Canada Dream Team that focuses on brain tumours that have the worst outcomes, such as glioblastomas. By combining wide and deep expertise in research, clinical strengths, and unique partnerships, the researchers hope to accelerate new cures for some of the hardest-to-treat cancers.
Currently, the prognosis for glioblastoma remains very poor, with fewer than 10 per cent of patients surviving beyond five years. Current therapies are severely limited. While PRMT5 inhibition has been suggested previously as a way to target brain and other cancers, no one has tested this strategy in a large cohort of patient tumour-derived cells that have stem cell characteristics, cells that are at the roots of glioblastoma growth. "We used a different strategy to stop cancer cells from proliferating and seeding new tumours," says Dr. Arrowsmith, who also leads the U of T site of the International Structural Genomics Consortium, a public-private partnership with labs around the world, accelerating the discovery of new medicines through open science. "By inhibiting one protein, PRMT5, we were able to affect a cascade of proteins involved in cell division and growth. The traditional way of stopping cell division has been to block one protein. This gives us a new premise for future development of novel, more precise therapies."
Recommended article
News • Deep learning applied to MRI scans
Glioblastoma: Using AI to improve prognosis and treatment
In the first study of its kind in cancer, researchers have applied artificial intelligence to measure the amount of muscle in patients with brain tumours to help improve prognosis and treatment.
"This strategy also has the opportunity to overcome the genetic variability seen in these tumours," says Dr. Dirks, who led the pan-Canadian team. "By targeting processes involved in every patient tumour, which are also essential for the tumour stem cell survival, we side-step the challenges of individual patient tumour variability to finding potentially more broadly applicable therapies."
The poor outcome of glioblastoma could be attributed, at least in part, to the presence of tumour-initiating cancer stem cells, which have been shown by the Dirks laboratory to drive tumour growth and resistance to therapy. Novel therapeutic strategies are urgently needed to target these cancer stem cells, in addition to bulk tumour cells.
Right now, we have too few medicines to choose from to make precision medicine a reality for many patients. We need basic research to better understand the mechanism of action of drugs, particularly in the context of patient samples
Cheryl Arrowsmith
In this study, researchers tested a group of new experimental small molecules designed to specifically inhibit key cellular enzymes being developed and studied by the Structural Genomics Consortium to see if any would stop the growth of glioblastoma brain tumour cells in the laboratory. The brain tumour cells were isolated from patients' tumours and grown in the laboratory in a way that preserved the unique properties of cancer stem cells. They found that specific molecules – precursors to actual therapeutic drugs – inhibited the same enzyme, PRMT5, stopping the growth of a large portion of these patient-derived cancer stem cells. Many current drugs do not eliminate cancer stem cells, which may be why many cancers regrow after treatment. But they also caution that actual treatments for patients are many years away, and require development and testing of clinically appropriate and safe versions of PRMT5 inhibitors that can access the brain.
The researchers also examined the molecular features of the patient-derived glioblastoma cells by comparing those that responded well to those that did not respond as well. They found a different molecular signature for the tumour cells that responded. In the future, this could lead to specific tumour biomarkers, which could help in identifying those patients who will respond best to this new class of drugs. "Right now, we have too few medicines to choose from to make precision medicine a reality for many patients," says Dr. Arrowsmith. "We need basic research to better understand the mechanism of action of drugs, particularly in the context of patient samples. This is what will help us develop the right drugs to give to the right patients to treat their specific tumours."
Source: University Health Network
15.02.2021