Article • On the way to diagnostic mainstream
Liquid biopsy to advance cancer diagnosis
Liquid biopsy offers a new dimension to detection and stratification of cancer – yet the technique also faces hurdles and challenges in becoming a mainstream diagnostic approach that will help facilitate more personalised treatments.
Report: Mark Nicholls
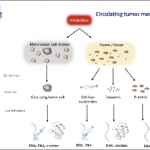
A critical challenge lies in identifying the extremely low concentrations of the bio-analytes of CTC (circulating tumour cells), ctDNA (circulating tumour DNA) and exosomes in the blood, explained Professor Klaus Pantel, Director of the Institute of Tumour Biology at the Hamburg Eppendorf University Medical Centre in Germany.

He believes, however, that this is an evolving technique which will offer benefits to patients and healthcare systems in terms of better diagnosis, targeted treatments and cost effectiveness. The professor outlined the ‘current clinical application and challenges’ at a recent conference held in Corfu, which looked at liquid biopsy advances.
Speaking to Healthcare in Europe after the meeting, he said a prime clinical application of liquid biopsy is in screening and early diagnosis of cancer ‘with the goal to find small tumours early enough to cure them by surgery’. However, he added: ‘We have to develop and use extremely sensitive methods because, to detect small tumours, we need to detect very small amounts of circulating DNA or circulating tumour cells or EVs (extracellular vesicles).’ He explained how good sensitivity and specificity are needed to detect tumour types but the approach can require a combination of different biomarkers (e.g., circulating DNA and proteins) to achieve high accuracy.
Recommended article
Article • Mass Spectrometry
Circulating biomarkers – the great white hope of liquid biopsy
About six years ago, liquid biopsy appeared on the diagnostic lab stage. Last December, a seminar offered by Agena Bioscience in Frankfurt, Germany, was set to explore the enormous potential of this new technique for oncology.
A second application of liquid biopsy can be to assess individual risk in cancer patients. ‘This can be done with circulating tumour cells and new data with circulating DNA that helps to subdivide patients into different risk categories beyond usual TNM staging,’ he said. ‘We can also use blood sampling to follow up patients during the post-surgical period and see whether we see any early molecular relapse detected in the blood, before the imaging tells us there is metastases.’
Pantel said the process can be honed to determine the therapy offered to patients – both those who are not fully metastatic and already have adjuvant or neo-adjuvant therapy, or those with full-blown metastases that can be seen from imaging analysis.
Ultrasensitive assays that enable the reliable detection of minute numbers of circulating tumour cells, or amounts of ctDNA, should be implemented into clinical trials of neoadjuvant and/or adjuvant therapies, he added, and might also complement imaging procedures currently used for post-surgical surveillance of tumour relapse. CTCs or ctDNA can also help identify potential druggable alterations on the DNA or druggable pathways.
The tumour is only a small part of the total body and the signal that comes from the tumour is also just a small signal in the blood so that is really the challenge
Klaus Pantel
Examples for druggable genetic alterations are the mutations in the EGFR gene in lung cancer and for druggable alterations that are the variant expression of androgen receptor in prostate cancer. ‘These are very good examples of using liquid biopsy analysis to better say which individual therapy works in individual patients,’ Pantel explained.
However, he said the major challenge remains in the fact that the bio-analytes of CTC and ctDNA are in such low concentrations in the blood. ‘The tumour is only a small part of the total body and the signal that comes from the tumour is also just a small signal in the blood so that is really the challenge,’ he continued. With smaller tumours, the challenge is to subtract the signal from the noise. Additionally, in older patients, there are mutations that do not necessarily lead to cancer, so there is background noise too.
He stressed the importance of testing the robustness and reproducibility of these methods, in particular with low DNA concentration from the tumour. Such work is currently being undertaken by the European Liquid Biopsy Society (ELBS) and other bodies.
Image source: Shutterstock/Mega Pixel
Latest innovations include developing methods with in-built controls, while in the CTC field advances are in measuring tumour cells in vivo. Wearables, he suggested, will play a role with the advent of devices that, within the next five years, will continuously measure and collect CTCs, addressing the problem of low concentration. While this lies in the future, there are already clear benefits from liquid biopsy for patients and healthcare systems. ‘If we can better stratify patients in terms of individual need for an expensive cancer therapy, we will save money because we will not waste expensive therapies on the wrong patients,’ Pantel added. ‘Using this technology to better define which patients may profit from it has a benefit both for the patient and the healthcare system.’
He concludes that liquid biopsies of blood samples for quantitative and qualitative assessments of CTCs and ctDNA can provide new insights into the disease stages in cancer patients and lead to more targeted treatments.
Profile:
Klaus Pantel is Professor of Medicine at the University Medical Centre Hamburg Eppendorf, Hamburg, in Germany and Director of the Institute of Tumour Biology as well as a member of the executive board of the university’s comprehensive cancer centre. He is also Coordinator and Director of the European Liquid Biopsy Society.
20.02.2020





