Article • Mass Spectrometry
Circulating biomarkers – the great white hope of liquid biopsy
About six years ago, liquid biopsy appeared on the diagnostic lab stage. Last December, a seminar offered by Agena Bioscience in Frankfurt, Germany, was set to explore the enormous potential of this new technique for oncology.
Report: Daniela Zimmermann and Walter Depner

European Hospital talked about the promise of liquid biopsy – and about its risks – with Professor Dr Klaus Pantel, Director of the Institute of Tumour Biology at University Hospital Hamburg-Eppendorf, a pioneer in liquid biopsy, and Professor Dr Jörg Kriegsmann, Medical Director at the Medical Service Center for Histology, Cytology and Molecular Diagnostics, Trier, Germany.
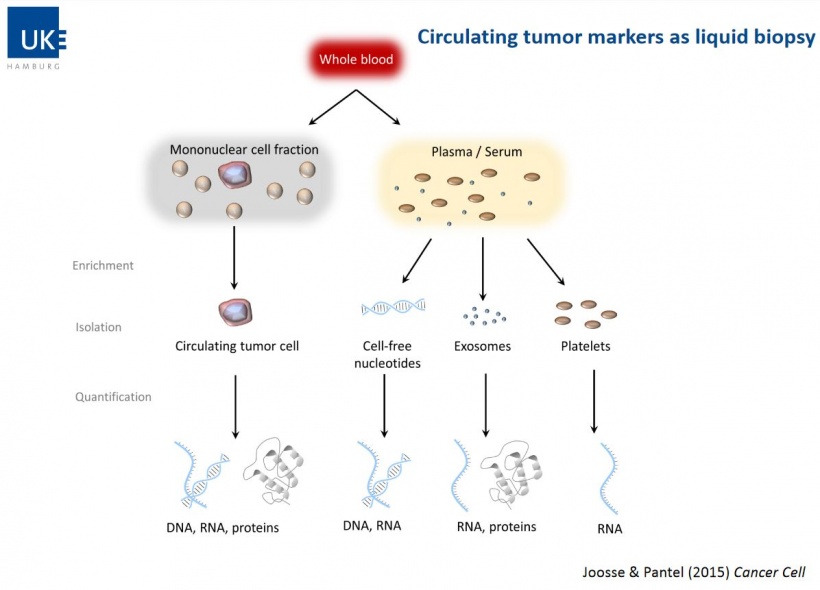
Liquid biopsy is a blood sample used to detect, characterize and monitor cancer non-invasively. A growing tumour always sheds dead cells containing DNA and releases them into body fluids. Once in the bloodstream, this circulating so-called cell-free DNA (cfDNA) can be isolated and amplified with PCR (polymerase chain reaction) and analysed with new molecular methods such as Next Generation Sequencing (NGS) or Agena’s Mass ARRAY technology which is based on Matrix-assisted laser desorption ionisation Time-of-flight (MALDI-TOF). While the fact that tumour cell fragments float in the blood has been known since the 1990s, modern genome analysis methods are required to exploit the potential of liquid biopsy and make this diagnostic procedure available for oncology.
In current cancer diagnostics tissue biopsy is the gold standard, albeit it has a major drawback: invasive sampling is risky for the patient. Moreover, certain regions, such as the brain or the lungs, are difficult to access with a biopsy needle and the interventions cannot easily and immediately be repeated. Liquid biopsy samples of circulating tumour cells (CTC) or circulating cell-free tumour DNA (cfDNA) in the blood are easy to obtain, and after thorough validation, can complement the invasive tissues biopsy.
Professor Pantel explains the hopes and objectives associated with liquid biopsy: "In oncology, our major aim is to detect a tumour as early as possible since early detection immensely increases the chances to cure the cancer. What we envision in fact is screening every person from a certain age for all major tumour types in order to be able to react quickly. Furthermore we are interested in issues such as cancer progression and the development of metastases.”
For Professor Pantel’s oncology team, as for research teams in many other clinical disciplines, therapy monitoring plays a crucial role. Cancer therapies are stressful for the patient and incredibly expensive; patients as well as the healthcare system would benefit from discontinuing unsuccessful treatments. Research on therapeutic response includes the precise identification of the target groups for certain therapies and aims to obtain insights into resistance mechanisms – where and in which patients do they develop?
In view of these objectives R&D today is looking for liquid biopsy tests with a broad range of applications. First-generation tests are commercially available and are being used successfully. But Professor Pantel warns of exaggerated optimism. He is sure it will take a while before the “ideal” blood test is found: “We have to aim for particularly high specificity because the older we get the more mutations our cells develop. Most of them are entirely harmless and won’t lead to cancer, even when they are found on tumour cells. False-positives are especially vicious as they mean we see indications of cancer cells where there is no reason for concern. At the same time certain tumour types cannot yet be identified with the minute traces we find.”

Professor Kriegsmann focused his presentation on cell-free tumour DNA (cfDNA). The concentration of cfDNA in cell fragments is increased in the presence of certain diseases, including certain types of cancer. Thus, cfDNA is used as a diagnostic biomarker.
Tumour composition is heterogeneous.This is why a standard tissue biopsy cannot detect aggressive tumour clones. Liquid biopsy finds DNA molecules of the entire tumour. It might thus be a valuable complement of tissues biopsy. cfDNA also provides important information in prenatal diagnostics, in transplant medicine and with regard to autoimmune diseases.
Today, says Professor Kriegsmann, NGS has become part of the standard workup in mutation analyses. The Mass ARRAY technology, however is a sensitive and cost-effective alternative to NGS in complex analyses of clinically relevant mutations. With its high sensitivity, robustness and quick availability of final this method is very well suited for clinical practice. In addition, it is easy to perform and requires little “hands-on time” compared to NGS.
At the seminar, Professor Kriegsmann presented impressive images to illustrate the progress that has been achieved in the field. But he also addressed the problems and limitations of liquid biopsy, for example when “false readings” can happen. Promising studies demonstrated the ability of liquid biopsy to quickly and reliably detect resistance mutation in lung cancer. Liquid biopsy further provides sensitive and fast analysis of KRAS and BRAF mutations which in turn allows the design of individualised therapies for patients with colorectal cancer.
“The availability of a new and highly sensitive DNA analysis method enables oncology researchers to chart entirely new analytics territory. Liquid biopsy, particularly when used to detect cfDNA, shows immense potential to complement the diagnosis of primary tumours. Today there is no standardized comprehensive quality management that covers all steps from sampling to sample processing and reading the results – which is an essential component of all analytic methods used for diagnostic purposes. Due to the easy availability of blood and the automation potential of the analytic process, liquid biopsy however can be a valuable complement to standardized histopathology methods. It seems to be a very attractive method, particularly in view of the fact that no tissue samples need to be obtained,” Professor Kriegsmann concluded.
Profiles:
Prof Klaus Pantel is Chairman of the Institute of Tumour Biology at the University Medical Center Hamburg-Eppendorf. The institute is part of the Centre of Experimental Medicine and the University Cancer Center Hamburg (UCCH). The pioneering work of Prof Pantel in the field of cancer micrometastasis, circulating tumor cells and circulating nucleic acids (ctDNA, microRNAs) is reflected by more than 400 publications in excellent high ranking biomedical and scientific journals. Moreover, Prof Pantel coordinates the European TRANSCAN group “CTC-SCAN”, the European IMI consortium CANCER-ID (www.cancer-id.eu) on blood-based “Liquid Biopsies” and serves on the Editorial Boards of international cancer journals (e.g., Clin. Cancer Res., Breast Cancer Res., Cancer Res.).
Professor Jörg Kriegsmann MD,Dr. phil. a specialist in Pathology, is co-founder and head of the Medical Supply Centre for Histology, Cytology and Molecular Diagnostics in Trier, and co-founder and partner of the cross-regional professional association for histology, cytology and molecular diagnostics in Trier, Düren and Düsseldorf.
30.03.2017






