Image source: Horiba
News • Analytical performance assessment
Assay for rapid D-dimer quantification validated in independent study
In a new study, scientists from the Hematology Department, University Hospital of Clermont-Ferrand, have successfully evaluated the analytical performance of the Yumizen G DDi 2 hemostatic immunoassay.
The assay, developed by Horiba Medical, was able to detect venous thromboembolism (VTE) with excellent accuracy, reliability and turnaround time on the manufacturer's Yumizen G800 fully automated hemostasis analyzer, the company reports.
The evaluation results are now published in Research and Practice in Thrombosis and Haemostasis1 and will also be presented at the forthcoming ISTH Congress 2022.
D-dimer measurement is one of the most commonly requested tests in the hemostasis laboratory and is frequently used to exclude VTE. While a number of different immunoturbidimetric assays for D-dimer are marketed for routine practice, their reliability and analytical performance can vary. The independent study compared the performance of Horiba Medical’s Yumizen G DDi 2 assay, designed for its Yumizen G hemostasis analyzer range, versus other available immunoturbidimetric D-dimer assays.
The study analyzed the precision of Yumizen G DDi 2 by running repeat assays with a standardized control sample and found its coefficient of variation to fall within an ideal range for diagnostic purposes. The lower and upper detection limits of the Yumizen G DDi 2 assay were comparable to other D-dimer immunoassays tested, and notably it excelled in assay linearity, which was found to be maintained for a broad range of concentrations. The calculated linear range (up to 32700ng/mL FEU) extended significantly further than that specified by Horiba Medical (up to 20000ng/mL FEU). This makes the Yumizen G DDi 2 assay ideal for the direct measurement of most clinical samples without need for manual dilution steps, therefore decreasing sample turnaround time, the manufacturer states.
D-dimer immunoassays can often be marred by interference from hemolysis, icterus and lipemia, or heterophilic antibodies such as HAMAs and RF, and the study evaluated the Yumizen G DDi 2 assay for each. No significant impact of hemolysis was found for hemoglobin concentrations up to 10g/L, which was greater than other D-dimer assays. In addition, no interference of HAMAs (up to 500ng/mL) and RF (up to 935UI/mL) was detected. Since RF levels rarely exceed 1000UI/mL in clinical practice,2 the safety of the Yumizen G DDi 2 assay was strengthened further.
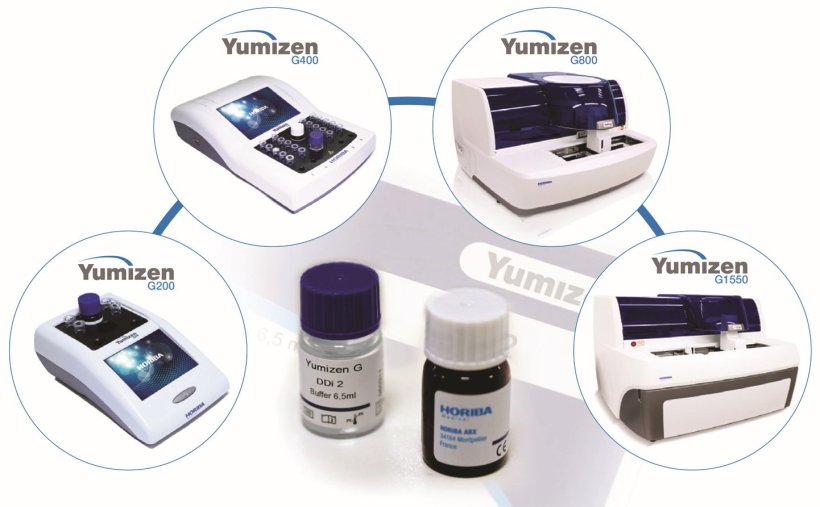
Horiba Medical’s Yumizen G DDi reagent kit is available for its comprehensive range of Yumizen G hemostasis instruments which covers the needs of any laboratory. The dedicated coagulation portfolio includes the fully automatic Yumizen G800, Yumizen G1500 and Yumizen G1550, as well as semi-automatic Yumizen G200 and Yumizen G400 analyzers. Using antibody-coated latex particles and measured via the immunoturbidimetric channels of these Yumizen G hemostasis instruments, Horiba Medical’s reagent kit delivers D-dimer results consistency and analytical performance reliability as demonstrated by the recent evaluation study.
References:
Source: Horiba Medical
04.05.2022