News • COVID-19 in radiology
CT outperforms lab diagnosis for coronavirus infection
Study from China shows computed tomography of chest demonstrates better sensitivity than RT-PCR technique
In a study of more than 1,000 patients published in Radiology, chest CT outperformed lab testing in the diagnosis of 2019 novel coronavirus disease (COVID-19). Researchers at Tongji Hospital in Wuhan, China, concluded that CT should be used as the primary screening tool for COVID-19.
In the absence of specific therapeutic drugs or vaccines for COVID-19, it is essential to detect the disease at an early stage and immediately isolate an infected patient from the healthy population. According to the latest guidelines published by the Chinese government, the diagnosis of COVID-19 must be confirmed by reverse-transcription polymerase chain reaction (RT-PCR) or gene sequencing for respiratory or blood specimens, as the key indicator for hospitalization. However, with limitations of sample collection and transportation, as well as kit performance, the total positive rate of RT-PCR for throat swab samples has been reported to be about 30% to 60% at initial presentation.
Compared to RT-PCR, chest CT imaging may be a more reliable, practical and rapid method to diagnose and assess COVID-19, especially in the epidemic area
In the current public health emergency, the low sensitivity of RT-PCR implies that a large number of COVID-19 patients won’t be identified quickly and may not receive appropriate treatment. In addition, given the highly contagious nature of the virus, they carry a risk of infecting a larger population. “Early diagnosis of COVID-19 is crucial for disease treatment and control. Compared to RT-PCR, chest CT imaging may be a more reliable, practical and rapid method to diagnose and assess COVID-19, especially in the epidemic area,” the authors wrote.
Recent research found that the sensitivity of CT for COVID-19 infection was 98% compared to RT-PCR sensitivity of 71%. For the current study, researchers set out to investigate the diagnostic value and consistency of chest CT imaging in comparison to RT-PCR assay in COVID-19.
Included in the study were 1,014 patients who underwent both chest CT and RT-PCR tests between January 6 and February 6, 2020. With RT-PCR as reference standard, the performance of chest CT in diagnosing COVID-19 was assessed. For patients with multiple RT-PCR assays, the dynamic conversion of RT-PCR test results was also analyzed as compared with serial chest CT scans.
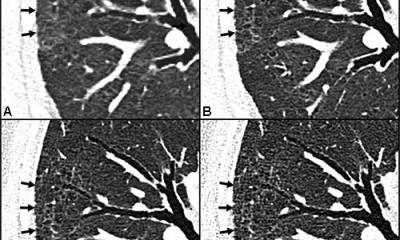
The results showed that 601 patients (59%) had positive RT-PCR results, and 888 (88%) had positive chest CT scans. The sensitivity of chest CT in suggesting COVID-19 was 97%, based on positive RT-PCR results. In patients with negative RT-PCR results, 75% (308 of 413 patients) had positive chest CT findings. Of these, 48% were considered as highly likely cases, with 33% as probable cases. By analysis of serial RT-PCR assays and CT scans, the interval between the initial negative to positive RT-PCR results was 4 to 8 days. “About 81% of the patients with negative RT-PCR results but positive chest CT scans were re-classified as highly likely or probable cases with COVID-19, by the comprehensive analysis of clinical symptoms, typical CT manifestations and dynamic CT follow-ups,” the authors wrote.
Source: Radiological Society of North America (RSNA)
28.02.2020