Image source: Inspirata
News • Software solution
Using AI to match cancer patients to early phase clinical trials
Cancer informatics and digital pathology provider Inspirata announced that King’s Health Partners ECMC and Guy’s and St Thomas’ NHS Foundation Trust will pilot its Trial Navigator software as part of an evaluation the organisations are conducting into how artificial intelligence based automation can improve the identification and efficiency of matching patients with cancer to early phase clinical trials.
Trial Navigator’s introduction as part of a pilot evaluation within the Cancer Early Phase Trials Unit will see Inspirata collaborate with both King’s Health Partners and the Experimental Cancer Medicine Centre (ECMC) Network Programme Office.
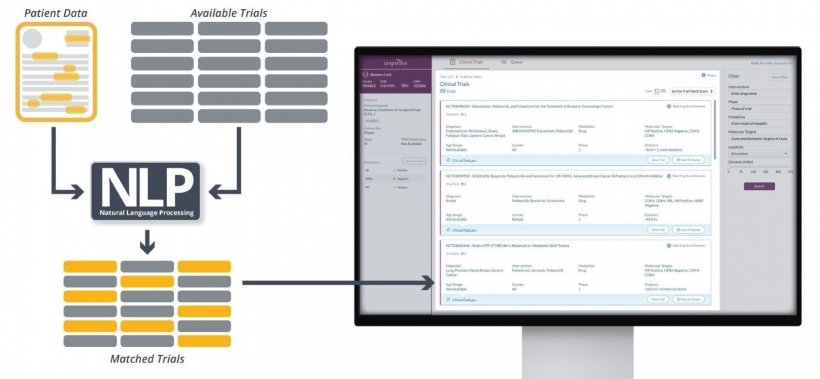
Delays in candidate identification and the absence of real-time visibility of open studies introduces a risk that patients miss out on trials for which they could have ultimately proved eligible. By applying oncology-specific natural language processing (NLP) to interrogate both the patient’s individual medical reports and potential trial eligibility criteria, this project will explore the extent to which Trial Navigator can help to improve bottlenecks in both identifying and matching patients to relevant clinical trials. “At the Cancer Centre at Guy’s, we’re constantly striving to get the best possible outcomes for our patients, and sometimes that means providing them with the opportunity to enrol on trials of new cancer drugs when conventional treatments have been unsuccessful. AI technology offers the potential to better match our patients to available trials, but we need to evaluate them first to prove that they can deliver on their promise,” says Danny Ruta, Clinical Artificial Intelligence Lead, Guy’s and St Thomas’ NHS Foundation Trust. “We hope that the intelligent automation afforded by Trial Navigator will prove to be an effective solution for identifying greater numbers of our eligible cancer patient population for clinical trials.”
We are excited to support a project that seeks to aid clinicians in the complex and time-consuming task of matching patients to suitable trials
Michelle Mitchell
“Our patients are looking to us for assurances that all possible options and avenues associated with their care have been thoroughly evaluated,” says Debashis Sarker, Reader in Experimental Oncology in the School of Cancer and Pharmaceutical Studies of King’s College London, and Honorary Consultant in Medical Oncology at Guy’s and St Thomas’ NHS Foundation Trust. “I am hugely attracted to any toolset which serves to augment my own understanding of the different trial options available so that I can impart this confidence and where applicable, see more patients obtain prompt access to potential new treatments in a more timely manner.”
While building on an underlying oncology NLP engine that has already been successfully deployed at over 400+ hospitals and cancer centres worldwide, Trial Navigator has been purposely designed to improve clinical trial matching by supporting both the clinician at the point of care, and trial coordinators with patient identification. Trial Navigator can also be deployed to perform instantaneous lookups against any relevant trial database. “Digital technology will play a key role in ensuring that the UK remains one of the best places in the world to conduct experimental cancer medicine studies, so that our UK patients get access to novel therapies at the earliest opportunity. We are excited to support a project that seeks to aid clinicians in the complex and time-consuming task of matching patients to suitable trials,” says Michelle Mitchell, Chief Executive of Cancer Research UK, which co-funds the ECMC network.
“We could not be more excited nor proud to work with Guy’s and St Thomas’, Kings Health Partners and ECMC on this project,” says Oenone Duroe, General Manager, Inspirata Europe. “We look forward to collaborating closely with the team to validate the efficacy of AI in a clinical trial matching context, and by drawing on insights derived, support equivalent Trial Navigator engagements within the National Health Service.”
Source: Inspirata
27.07.2021





