Image credit: Franciszek Lukaszczyk Oncology Center; Harat et al., Nature Communications 2023 (CC BY 4.0)
News • Glioma imaging
PET imaging for more effective treatment of aggressive brain tumors
A more refined approach to the precise removal of brain tumors is on the horizon.
The long-anticipated advancement, made achievable through the adaptation of tumor imaging processes employing positron computed tomography, turns out to be possible. The recent findings, unveiled in the journal “Nature Communications”, are the result of collaborative efforts undertaken by healthcare and scientific institutions in Bydgoszcz, Poland. Noteworthy contributions have been made by the personnel of the Franciszek Lukaszczyk Oncology Center (CO), 10th Military Research Hospital, Collegium Medicum of the Nicolaus Copernicus University (CM UMK), and Bydgoszcz University of Science and Technology (PBS). These investigations hold the potential to serve as the cornerstone for revising the established norms in the treatment of gliomas.
To achieve optimal tumor removal, it is imperative that a neurosurgeon or radiation oncologist possesses the utmost precision in identifying the spatial distribution of glioma cells within the patient's cerebral tissue. At present, this crucial information is primarily acquired through the use of nuclear magnetic resonance imaging (MRI). During surgical interventions, the neurosurgeon removes the extent of the malignant lesion as visualized in the MRI scans, while, during radiotherapy, a standard safety margin is employed, extending approximately two centimeters beyond the observed tumor boundaries.
With the anticipated integration of this method into clinical practice, there is a hopeful prospect of improving the treatment outcomes associated with one of the most challenging malignancies among human cancers
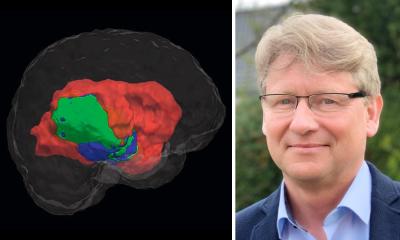
Maciej Harat
“In the course of our study, we have demonstrated our ability to assess high-grade glioma infiltration beyond the observable boundaries provided by contrast-enhancement in magnetic resonance imaging. This suggests that even the most aggressive tumor resection guided by MRI imaging may inadvertently leave a significant portion of the tumor unaddressed, consequently precipitating rapid disease recurrence. This discovery is a direct outcome of our principal achievement, namely, the validation of an alternative brain imaging modality: positron emission tomography utilizing fluorine-labeled tyrosine,” says Prof. Maciej Harat (CO, PBS), who is not only one of the architects of the research framework but also the primary author of the scholarly article unveiling the discovery.
Positron emission tomography (PET) imaging of gliomas begins with the administration of safe doses of the rapidly decaying radioisotope fluorine-18 to the patient. This isotope is incorporated into tyrosine molecules, an amino acid found in considerably higher concentrations within cancerous cells than in the healthy brain tissue. When the tyrosine molecule is situated within the tumor, and the radioisotope atom decays, one of the resultant emitted particles is a positron – a positively charged antiparticle corresponding to the electron. Given the presence of numerous electrons within the patient's body, the positron promptly interacts with an electron, leading to mutual annihilation and the conversion of their mass into energy. As a result of this annihilation, two photons possessing distinct energies are emitted in opposite directions. A PET tomographic apparatus detects these photon pairs and, by discerning the disparities in their detection times, establishes the precise location where annihilation transpired. This process serves as a reliable indicator of the location of tumor cells.
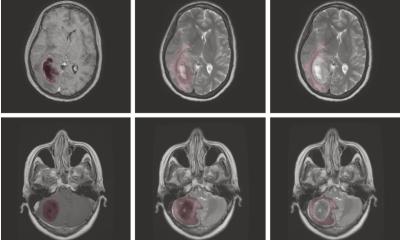
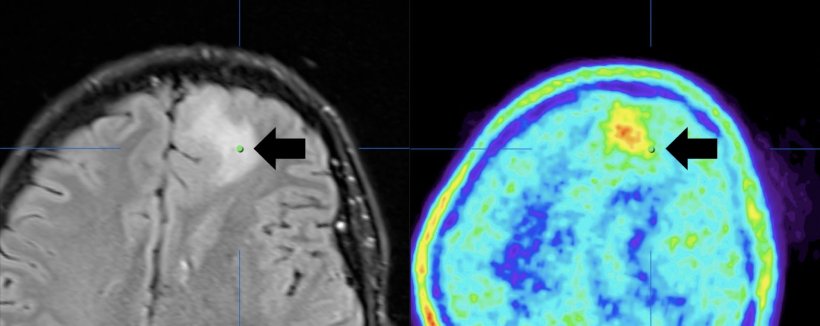
Previous research conducted by Professors Maciej Harat and Bogdan Malkowski (CM UMK) has shed light on a promising approach to glioma imaging that involves complementing MRI with PET using tyrosine and conducted at two distinct time points. Typically, attempts to utilize tyrosine PET as a marker for defining the boundaries of malignant gliomas occurred approximately 20 to 40 minutes following the administration of the radiotracer. However, a pivotal supposition put forth by Prof. Malkowski was that tyrosine molecules might exhibit earlier uptake by the tumor – a hypothesis that was swiftly validated. When the PET examination was performed shortly after the tracer's administration, the resulting glioma image diverged substantially, owing to the pronounced tyrosine uptake in the most aggressive tumor cells. Additionally, it became evident that the glioma image obtained through this method differed significantly from those obtained via MRI or conventional PET imaging techniques. Notably, this innovative approach revealed instances where disease progression became observable up to eight months earlier compared to traditional MRI methods.
“In the field of neuro-oncology, positron emission tomography is progressively gaining greater application in diagnostic and disease monitoring procedures. However, magnetic resonance imaging continues to play an important role in precisely defining the surgical and radiotherapeutic targets. To substantiate the enhanced utility of PET employing tyrosine, our study necessitated not only to verify the cellular characteristics in regions where the imaging outcomes of both methods diverged substantially but also to demonstrate that it is in the early post-tyrosine administration phase that we identify the most aggressive areas of malignant tumors,” explains Prof. Maciej Harat.
The key rationale behind the proposed refinement in glioma imaging finds its basis in a structured series of serial brain stereotactic biopsies. Building upon PET imaging conducted at the Oncology Center in Bydgoszcz, brain tumors were biopsied at the 10th Military Research Hospital. Following the patients' consent, small tissue samples were extracted from regions exhibiting alterations in both PET and/or MRI scans. The exhaustive analysis of the biological material validated the considerably greater accuracy of the new glioma imaging method. This breakthrough bears significant implications for patients, as it promises not only more precise eradication of cancerous cells during surgical interventions or radiotherapy but also the preservation of healthy brain tissue. “The amassed evidence will serve as a foundation for more comprehensive surgery or radiotherapy planning in subsequent clinical trials. With the anticipated integration of this method into clinical practice, there is a hopeful prospect of improving the treatment outcomes associated with one of the most challenging malignancies among human cancers,” emphasizes Prof. Maciej Harat.
The development of a new glioblastoma imaging procedure stems from rigorous scientific research. Crucial clinical trials aimed at promoting the adoption of tyrosine-based imaging and, consequently, enhancing the efficacy of brain tumor treatment have progressed to an advanced stage. The preliminary findings align with the anticipated outcomes of the research community. It is, therefore, reasonable to expect that the presented imaging method will see widespread adoption in the coming years.
Source: Politechnika Bydgoska im Jana i Jedrzeja Sniadeckich
27.10.2023