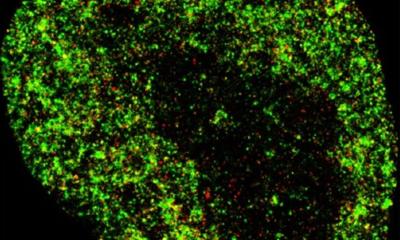
Image courtesy of Dominic Zerulla/PEARlabs
Article • Super-resolution miscroscopy
PEAR: setting nano-imaging in motion
Ever since the Abbe diffraction limit of conventional microscopy has been surpassed, super-resolution techniques have been diving ever deeper into the most miniscule details of molecular structures. We spoke with Prof. Dominic Zerulla, whose company PEARlabs is developing an imaging technique that sets out to push the boundaries once more – by looking at in-vivo nano-scale processes in motion.
Report: Wolfgang Behrends
Resolution in light microscopy is limited by the light’s wavelength, which, at the short end of the spectrum, is around 380 nanometres (nm). Sophisticated equipment can distinguish objects with around half that size, anything smaller becomes a blur because the visual information does not transfer to the far-field (the region visible with the human eye). With the help of structured illumination and signal computation, this limit can be further reduced to slightly below 100 nm. While this might be just enough to make out the coronavirus SARS-CoV-2, finer details such as the peplomers on the virus surface that create the characteristic shape remain obscure at this magnification level. “Thus, to be able to understand what is going on, we need to go deeper,” says Zerulla.

How much deeper? In this case, at least an order of magnitude beyond anything conventional microscopy has to offer. While this can be achieved using super-resolution fluorescence microscopy, some benefits of the technique only apply in theory, the expert cautions: “To illuminate the target, synthetic or organic molecules – for example green fluorescent proteins (GFPs) – must be implemented into the genetic code. However, our current understanding of genetics is limited, so we cannot insert these molecules wherever we want – in some cases, it works, but in most, it doesn’t.”
Furthermore, the maximum spatial resolution depends on the effectively achievable signal-to-noise level: While a highly stable material such as diamond allows for very intense illumination and hence single nanometre details, any biological matter would simply be obliterated by the beam’s energy output.
Extremely fast & incredibly close
To get close enough to their target without destroying it, the research team opted for a different approach they named plasmonics electronically addressable super-resolution – in short, PEAR. Using an optical chip, the technique captures optically excited plasmonic resonances that are modulated when a minuscule metal plate is subjected to an electric current. By utilizing different frequencies, the chip carries over sub-diffraction limit visual information into the visible far-field. “Think of the metal plate as a single pixel. When you arrange multiple plates on an array, you can assign different frequencies to the individual plates. That way, an image can be generated,” Zerulla explains.
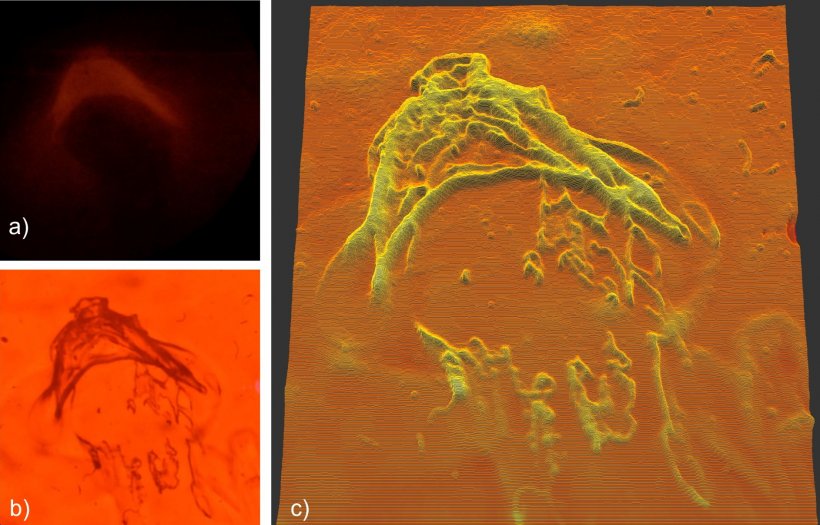
Image courtesy of Dominic Zerulla/PEARlabs
This method brings several benefits: Samples can be viewed without labelling dyes or exposing them to intense heat, vacuum, or other unsuitable environments. Combined with the ability to interrogate multiple ‘pixels’ simultaneously in high frequency, this leads to real-time observation of in-vivo samples. “Processes are much harder to understand using only still images – showing them in a video opens up a lot more possibilities.”
The current prototype configuration delivers detailed images via scanning with arrays consisting of 36 pixels at 10 frames per second (fps). “With enough computing power, we can generate nanometre-scale videos with much higher resolutions at very high frame rates of 200 fps,” says Zerulla. Magnification is limited by the size of the individual ‘pixels’; current manufacturing techniques achieve results in the 20 nm range, but Zerulla is confident that this will decrease due to advances in lithography. Using signal oversampling to bring this limit down even further, single-digit nanometre imaging with high frame rates should be entirely possible, he states. This could give detailed insights into the proliferation of cancer cells, viral infections, or drug delivery mechanisms and eliminate much of the trial-and-error in research that results from many biomedical processes being too small to see.
Currently, signal computing is a major bottleneck: “The PEAR technology can easily be upscaled to handle extremely high resolutions at frame rates well beyond the 30 to 60 fps the human eye can perceive,” says the PEARlabs CEO. “But this requires a lot of processing power, so we need a chip that can handle the information fast enough.” For this reason, the company is currently looking for investors to realise the full potential of the technique. “In the right setting, this may be the key to breakthrough discoveries,” Zerulla says. Further possible applications include mobile devices – chips in the fingerprint sensor of smartphones could be used for self-screening or point-of-care testing in remote locations.
Profile:
Dominic Zerulla is Associate Professor at the University College Dublin School of Physics. Following his studies in physics and a PhD in physical chemistry he headed the biophotonics group at Heinrich Heine University in Düsseldorf, Germany, where he received his habilitation in the same group that developed the first optical configuration for surface plasmon polariton excitation. Furthermore, he was a Visiting Professor for advanced optics at the Astrakhan State University, Russia, and Visiting Professor at the University of California at Berkeley. Prof. Zerulla is the founder and CEO of PEARlabs, a company which specializes in providing commercial solutions for super-resolution imaging using a novel electronically addressable photonic chip. He authored four book chapters, six patents, more than 120 articles, reports and letters in the fields of surface science, nano-optics, and plasmonics and he has given more than 110 talks at international conferences and universities.
30.12.2021