News • Ornithine Transcarbamylase deficiency
OTC deficiency: First patient benefits from gene therapy trial
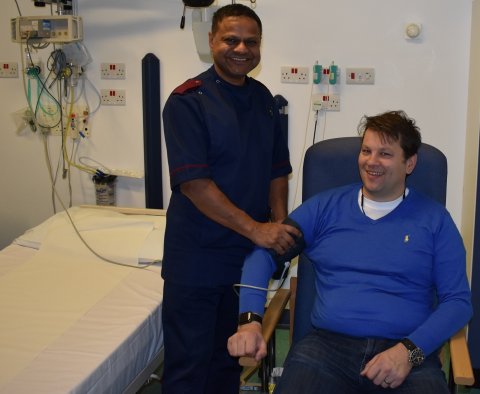
A patient at the Queen Elizabeth Hospital Birmingham (QEHB) was the first person in the world to take part in a pioneering gene therapy trial for Ornithine Transcarbamylase (OTC) deficiency, a rare disease that causes toxic levels of ammonia to build up in the blood.
Simon Smith, 45, was diagnosed with OTC deficiency as a teenager. Although he defied medical expectations by living a full life in his 20’s and 30’s, in recent years Simon’s health has taken a turn for the worse. This resulted in a referral to the QEHB Inherited Metabolic Disorders team. Dr Tarek Hiwot, Consultant in Inherited Metabolic Disorders, said: “QEHB is a specialist regional centre for rare diseases, meaning we are involved in ground-breaking clinical trials that have the potential to transform patients’ lives. “We have worked with Ultragenyx on a number of clinical trials, including the trial Simon has taken part in, which could potentially cure OTC deficiency.”
University Hospitals Birmingham NHS Foundation Trust, which QEHB is part of, is the only Trust in the country involved in the trial, which involves a single intravenous injection that targets the liver. The injection of a benign virus, DTX301, contains a treatment for OTC deficiency that helps the body to produce the correct proteins to help stabilise ammonia levels.
OTC deficiency has a number of symptoms, including an inability to stabilise ammonia levels in the body. This can cause issues with the liver, tiredness, and lead to delirium. Simon added: “Taking part in this trial has absolutely changed my life. I’ve gone from taking dozens of tablets a day to just three, and all my other symptoms seem to have gone. The care and support from Dr Hiwot and Research Charge Nurse Vishy Veeranna has been brilliant, and I can’t thank them enough. The trial has literally saved my life!”
This offers a potential new way to treat the disease that directly targets the genetic defect, rather than just treating the symptoms
Vishy Veeranna
Simon took part in the trial in August 2017 and has regular follow-up appointments at the NIHR/Welcome Trust Clinical Research Facility, on the QEHB site, to assess how effective the treatment has been. He was closely observed during and immediately after the transfusion, and has had weekly blood tests, which have monitored the effectiveness of the treatment and whether the body has rejected the treatment. “Over the last four years, research within the Inherited Metabolic Disorders team has grown massively, with over 140 patients with metabolic disorders or other rare disorders currently involved in clinical trials,” added Vishy. “The clinical trial Simon has taken part in uses a one-time intravenous infusion gene transfer to deliver a working copy of the OTC gene to the liver, offering a potential new way to treat the disease that directly targets the genetic defect, rather than just treating the symptoms. If successful – as seems to be the case for Simon – the liver cells will produce the OTC protein that is needed for a normal life.”
Although Simon’s sister carries the gene that causes OTC deficiency, she is less affected, with men more likely to be affected by the condition than women. The trial is continuing, and is currently recruiting patients to its third cohort from twelve centres in the world, based in the United States, Canada, Spain and UHB. The current standard treatments for OTC deficiency can include adhering to a strict low-protein, high-carbohydrate diet, daily medication, and in some cases dialysis to reduce high levels of ammonia in the blood.
Source: University Hospitals Birmingham NHS Foundation Trust
30.01.2019