Image source: Heo et al., Journal of Nuclear Medicine 2021
News • Abdominal aortic aneurysm
Novel radiotracer shows promise to predict AAA rupture
A new positron emission tomography (PET) radiotracer can detect abdominal aortic aneurysms (AAAs) and potentially predict when they will rupture, according to research presented at the Society of Nuclear Medicine and Molecular Imaging 2021 Annual Meeting.
Targeting a novel biomarker associated with AAA, the radiotracer is effective both in diagnosis and in providing information to assist in the development of AAA treatments, of which there currently are none.
The abstract in the Journal of Nuclear Medicine can be found here.
There is an unmet clinical need for a novel molecular biomarker to determine the underlying processes that lead to aneurysm expansion and rupture
Gyu Seong Heo
AAA is a life-threatening degenerative vascular disease. It occurs when blood vessels weaken and a bulge forms in the abdominal aorta (the vessel that supplies blood flow to the abdominal organs and the legs). AAAs typically remain asymptomatic until they rupture, leading to high mortality and a substantial burden on the health care system. “Right now, clinical diagnosis of AAA relies on anatomic measurements of AAA diameter, which is a poor marker for the prediction of rupture,” noted Gyu Seong Heo, PhD, a post-doctoral researcher at Washington University School of Medicine in St. Louis, Missouri. “Thus, there is an unmet clinical need for a novel molecular biomarker to determine the underlying processes that lead to aneurysm expansion and rupture and to serve as a therapeutic target for better management of AAA patients.”
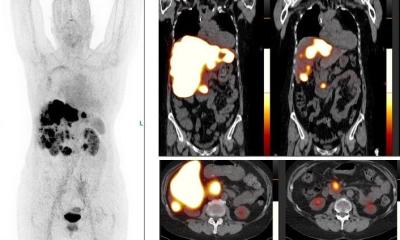
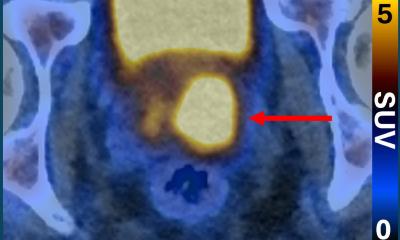
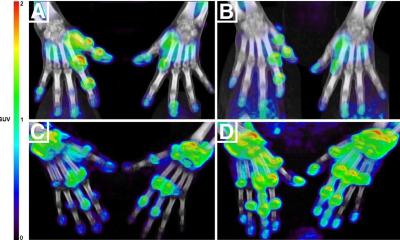
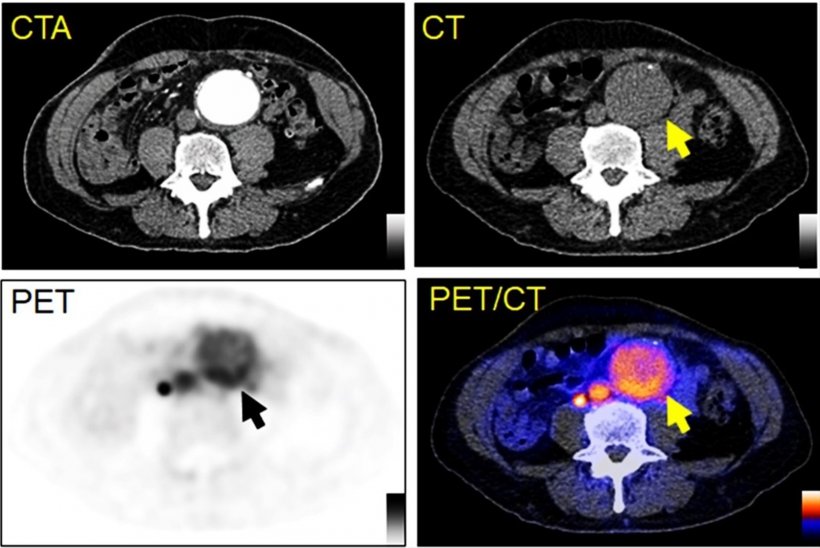
To explore this clinical need, researchers identified chemokine receptor type 2 (CCR2) as a potential, novel biomarker for AAA evaluation. They developed the novel PET tracer, 64Cu-DOTA-ECL1i, and it has been used to perform first-in-AAA patient imaging. 64Cu-DOTA-ECL1i PET was confirmed to be safe and effective for imaging CCR2 in AAA patients.
64Cu-DOTA-ECL1i was also utilized to assess CCR2-targeted treatment in preclinical animal AAA rupture models. In the models, 64Cu-DOTA-ECL1i imaging was highly suggestive of subsequent AAA rupture. Additionally, in a designated cohort of animals that received a CCR2 inhibitor as a form of therapy, researchers were able to demonstrate the effective prevention of AAA rupture. “Given the availability of CCR2 inhibitors for human uses, our work holds great potential to assess AAA vulnerability, screen AAA patients for CCR2-targeted treatment, and determine treatment response for optimal outcome,” said Heo.
Source: Society of Nuclear Medicine and Molecular Imaging
23.06.2021