© NEJM
Article • Over-detection and biopsies
New procedures for prostate cancer screening
Organised screening enabling early detection and treatment of prostate cancer can reduce death by an impressive 20%, based on 16 years of findings of over 160,000 men participating in the landmark European Randomised Study of Screening for Prostate Cancer (ERSPC).
Report: Cynthia E. Keen
However, the screening process, which includes prostate-specific antigen (PSA) testing followed by transrectal ultrasound-guided biopsy of men with elevated PSA levels of 3 ngml and higher, is responsible for overdiagnosis and excessive biopsies of low-grade prostate cancer. In fact, the World Health Organisation (WHO) Region Europe does not recommend systematic screening, citing a “high harm/benefit ratio”.

© Danderyd Hospital, Stockholm, Sweden
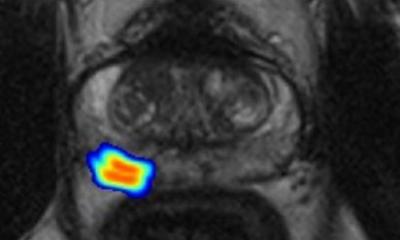
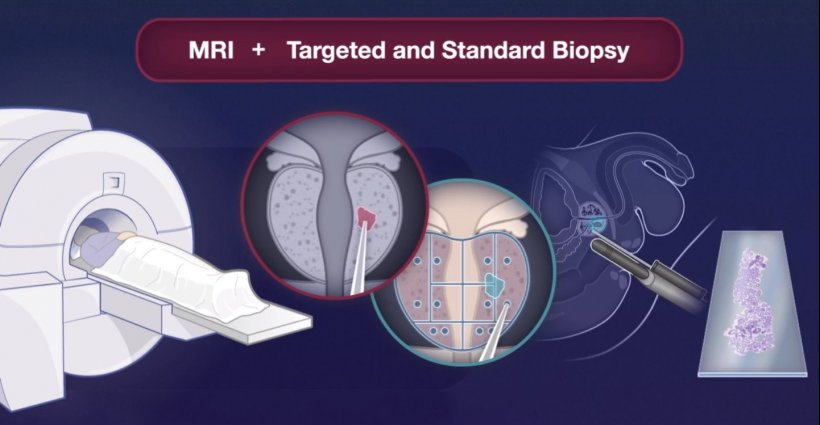
Important findings from a randomised clinical study of 12,750 men living in Stockholm County, Sweden, reported this summer at the European Association of Urology annual congress, may change this. Researchers from Karolinska Institute in Stockholm reported that MRI-guided biopsies can reduce overdiagnosis by half compared to traditional ultrasound-guided biopsies. The additional use of a novel blood test -- the Stockholm3 test (A3P Biomedical AB, Stockholm) -- reduced the number of MRIs performed by 36%, while further preventing the need to biopsy minor, low-risk tumours by 8%. ‘Our results show that we have identified the tools needed to carry out effective and safe screening for prostate cancer,’ said Tobias Nordström MD PhD, principal investigator of the STHLM3MRI study. ‘After many years of debate and research, it feels fantastic to be able to present knowledge that can improve healthcare for men.’
Prostate cancer is the third highest cause of cancer deaths in the European Union, with an estimated 78,800 men dying in 2020. Globally, it is the sixth leading cause of cancer death among men, with an estimated 359,000 deaths in 2018 based on the GLOBOCAN database, and is estimated to increase to 740,000 deaths by 2040 due to an aging population.
Recommended article
News • Stockholm3 test to reduce MRI
New blood test to improve prostate cancer screening
Researchers at Karolinska Institutet recently reported that magnetic resonance imaging (MRI) could reduce overdiagnoses and thereby improve prostate cancer screening. Now, the same research group has published a study in The Lancet Oncology, which shows that the addition of a novel blood test, the Stockholm3 test, can reduce the number of MRIs performed by a third while further preventing the…
The clinical study
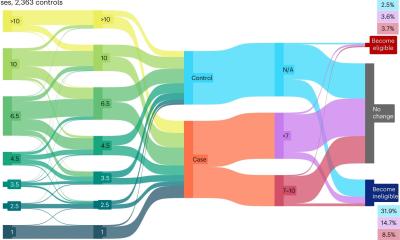
For the clinical study, 49,118 men aged 50-74 living in Stockholm County were randomly selected and invited to participate in a prostate cancer screening to identify whether they were at elevated risk of developing prostate cancer. Between 2018 and 2020,12,750 accepted and provided blood specimens; 2,293 with elevated risk as assessed by PSA and the Stockholm 3 test were randomly assigned to either a group who had traditional biopsies performed or a group having a MRI exam followed by a targeted biopsy approach in men with visible lesions on the MRI.
The Stockholm3 test uses an algorithm to analyse a combination of protein markers, genetic markers, and clinical data. Specifically, it determines the concentration of five plasma markers in a blood sample, as well as the analysis of approximately 100 genetic variants. The results of the analysis are integrated into an algorithm that includes the patient’s age, family history, and the result of a previous biopsy, if any. This creates a risk percentage score that a prostate biopsy will have a Gleason score of 7 or greater. ‘The combination of improved risk prediction and MRI-targeted biopsy has the potential to establish a new framework for prostate cancer screening,’ the researchers said.
An innovation such as the STHLM3MRI study makes an even more compelling case for the European Commission to ensure that a risk stratified approach to early detection of prostate cancer is adopted across the whole of Europe
Hendrick Van Poppel
The use of MRI-guided and targeted biopsies proved as effective as standard biopsies. The benefits of MRI are that it can be used to discriminate the need for a biopsy based on the presence/absence of suspicious lesions, and then to target biopsies to those lesions. The researchers said that, in a population of 10,000 men with elevated PSA levels, the MRI approach would result in 409 fewer men having a biopsy, 366 fewer biopsies with benign findings, and 88 fewer clinically insignificant cancers detected than with the standard biopsy approach. ‘Our clinical study showed that the Stockholm3 test can significantly reduce the number of MRI biopsies, without compromising the diagnostic capacity of medium grade prostate cancers, in comparison with the use of the PSA value 3 ng/mL as the cut-off value for biopsy recommendation,’ they said.
Professor Hendrick Van Poppel, Adjunct Secretary General of the EAU, commented, ‘It’s exciting to see breakthroughs such as this in the field of early detection of prostate cancer. An innovation such as the STHLM3MRI study makes an even more compelling case for the European Commission to ensure that a risk stratified approach to early detection of prostate cancer is adopted across the whole of Europe. The EAU is working hard to ensure that early detection is addressed in the implementation of Europe’s Beating Cancer Plan in order to reduce mortality of Europe’s most common male cancer while also dealing with the challenges of challenges of overdiagnosis and overtreatment.’
The researchers are currently performing cost-effectiveness analyses using data from the clinical trial. The Stockholm3 test is currently available in Sweden, Finland, Norway and Denmark.
Profile:
Associate professor of Urology at the Department of Clinical Sciences at Danderyd Hospital at Karolinska Institute, Tobias Nordström MD PhD combines clinical surgical practice with independent research. The latter focuses on development and implementing improvements in clinical care, such as targeted prostate biopsies and imaging and developing improved guidelines for treatment.
17.11.2021