© MohammedElAmine - stock.adobe.com
News • Microscopy
New microscopy method maps lipids without labels
A team at Helmholtz Munich and TUM has developed a technique that distinguishes lipid species in living cells without chemical labelling.
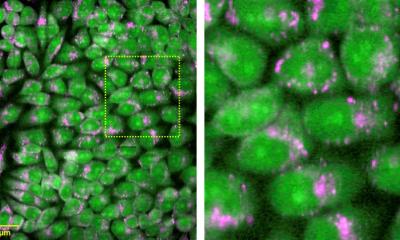
A team at Helmholtz Munich and the Technical University of Munich (TUM) has developed a new microscopy technique that can distinguish lipid species in living cells – in particular cholesterol and sphingomyelin – and map them without the need for chemical labelling. By combining mid-infrared illumination with optoacoustic detection, the method reads the lipids' natural spectral fingerprints, eliminating the need for specific fluorescent tags, which are laborious to develop and may interfere with lipid function. The team reports its results in the journal Nature Methods.
Lipids are key building blocks of cell membranes and help control how cells transmit signals and transport substances. However, sensing or visualising specific lipid classes in living cells is challenging. Traditional fluorescence microscopy requires developing custom fluorescent labels for each lipid – a time-consuming and costly process – and these labels can sometimes affect the lipid's function, stress the cells, or fail to bind specifically to the intended target.
Mid-infrared light and ultrasound make lipids visible without labels
A team led by Prof. Vasilis Ntziachristos, Head of the Bioengineering Center and Director of the Institute of Biological and Medical Imaging (IBMI) at Helmholtz Munich, as well as Professor and Director of the Chair of Biological Imaging at the Technical University of Munich, has tackled this challenge by developing a new microscopy method called hyperspectral mid-infrared optoacoustic microscopy (HyFOPM), which makes lipids visible without labels.
The method illuminates the sample with pulsed mid-infrared light at multiple distinct wavelengths, a so-called "hyperspectral" illumination. Lipids absorb specific bands of this light, producing a tiny, brief temperature rise that generates ultrasound waves. These waves are detected by ultrasound transducers and converted into a spectral image. Computational analysis of this spectral image then produces maps showing the distribution of different lipids within the sample.
The fingerprint region makes the difference
What makes this method unique is the wavelengths it uses. Many label-free approaches rely on spectral ranges dominated by bonds found in a wide variety of biomolecules, producing similar signals that make it hard to distinguish individual lipid classes.
The team instead focuses on the so-called fingerprint region, where absorption features arise predominantly from vibrational modes that are highly characteristic to each lipid's molecular structure. This means the method can detect not just the building blocks of a molecule, but also how they are arranged in relation to each other. By operating in this region, the technique can differentiate between lipid species – even chemically similar ones such as glycerophospholipids and sphingomyelin.
Minimal stress for cells
To test the reliability of these chemical fingerprints, the team compared their measurements with conventional infrared spectroscopy, which served as a reference. While the traditional method provides accurate measurements of dissolved compounds and solution droplets, it is not suitable for living cells, which require special preparation that can stress the cells.
The new microscopy technique not only reproduced the expected lipid spectra in solution but also enabled measurements directly in living cells. "A key advantage of HyFOPM is that label-free lipid observation results in minimal stress for living cells," says Francesca Gasparin.
Outlook: from cell culture to patient-adjacent applications
In the long term, the researchers see applications across a wide range of lipid research, from basic science to medical applications that have so far been difficult to access.
Source: Helmholtz Munich
04.05.2026