Source: Takeuchi et al., Biomedical Microdevices 2019
Article • Diabetes
Microneedles: Nano-sized, huge impact
Drug delivery, blood extraction, contrast agent injection – many procedures in modern medicine would be utterly impossible without needles. Despite the benefits, inserting pointy metal tubes into a patient also comes with several drawbacks. By downscaling the to micrometer-size, Japanese researchers open even more areas of application for needles, while bypassing some of the most important issues. We spoke with Prof. Beomjoon Kim, Director of the Laboratory for Integrated Micro Mechatronic Systems (LIMMS) at the University of Tokyo, about microneedles, the challenges for production and medical applications.
Report: Wolfgang Behrends
Using microneedles instead of their regular-sized counterparts minimizes the damage done to the tissue, which not only reduces pain, but also the risk of infection. However, manufacturing microneedles for medical purposes is a huge challenge because the needles need to fulfil several criteria: They must not exceed a certain size to be able to harness the capillary effect (e.g. to extract liquids from tissues).
At the same time, they need a high level of stability to withstand the penetration of human skin. Additionally, the needles must be biodegradable, so that debris left behind in the body (e.g. from tearing off a patch) do not cause problems within the patient’s body. This excludes the use of metal- or silicone-based materials. Instead, the LIMMS researchers propose using needles made of bio-compatible polymers with a porous, sponge-like structure.
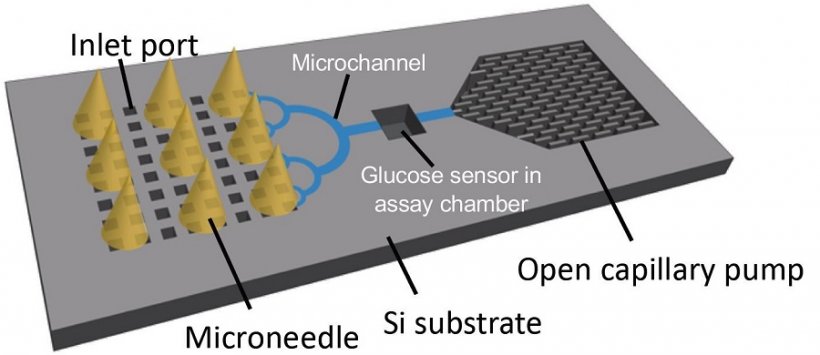
In their approach, the researchers arranged the microneedles on a paper sensor and an adhesive patch. When placed upon the skin, the biofluid is extracted via capillary force and transported to the sensor to detect, for example, blood glucose levels. A special dye on the paper then indicates the presence of the biomarker. “This process happens in less than 2 minutes”, explains Kim. “If a certain level of glucose is surpassed, the sensor changes color, so the result can be seen with the naked eye.”

Small scale leaves no room for error
Commercialisation of the technology has proved difficult so far. A major hurdle is adhering to minuscule dimensions while ensuring consistent needle shapes. “If even the slightest variance occurs, the patches will cause pain for the wearer,” says Kim. “There is basically no margin for error, as any structure larger than 30 micrometers will be detected when penetrating the skin.” This is an issue for current manufacturing processes such as micromolding and droplet-born air blowing (DAB).
The researchers succeeded in overcoming these challenges with a combination of advanced 3D printing and using a shadow mask to distribute the material with a higher degree of precision. Using a combination of additive manufacturing and wet shaping even allows for drug-coated microneedles with sufficient stability.

The first area of application is the early detection of prediabetes. Affixed to an adhesive patch, the needles extract interstitial fluid (ISF) onto a paper sensor, which checks the fluid for glucose. This could be paired with an automated insulin pump, which administers its dosage according to the glucose level monitored by the sensor. “Of course, the technology can be adapted to monitor other biomarkers, for example cholesterol,” Kim points out.
In light of the current pandemic, the team is working on a sensor patch to check for Coronavirus antibodies in the ISF. “One particular strength of the microneedles is the possibility of monitoring for an extended period of time, without causing discomfort to the wearer.” Much like a common plaster, the sensor patch can be applied by the patients themselves.
Needle design can be adapted for different uses
Instead of extracting liquids from the wearer, microneedle patches can also be designed for transdermal drug delivery. Depending on the type of pharmaceutical ingredient to be applied, the needles can be produced in a variety of shapes, lengths, and materials, while maintaining the quality standards for the needles, Kim stresses. Compared to the needles used for monitoring purposes, the drug-delivering structures require greater strength. However, due to the refined manufacturing method, their application is still painless.
The microneedle patch is currently in trial stage, but Kim estimates that it will be commercially available within the next 18 months. “We are working together with major medical device manufacturers in Japan, which greatly benefits the development process.”
Future plans

Looking ahead, the researchers at LIMMS are working to apply the technology to patches for continuous use. “These patches will be able to gather biomarkers from the ISF for about six hours,” Kim says. This will open up new areas of application for the tiny needles. Further plans include sensors with multiplex capacity, to monitor several biomarkers at once.
Prof. Kim also hopes to improve vaccination with the technology. “With our microneedles, we can facilitate distribution in developing countries, for example in Africa, because there is less need for cold-chain transportation.” This could be used to prevent spread of the Nipah virus, but also curb Covid-19 infections. “We aim to create a minimally invasive and painless self-diagnosis chip for early detection of the new coronavirus using a biodegradable and porous microneedle patch."
Profile:
Prof. Beomjoon KIM, Ph.D., is Director of the Laboratory for Integrated Micro Mechatronic Systems (LIMMS), a joint research unit of the University of Tokyo, Japan, and the French National Centre for Scientific Research (CNRS).
20.12.2021





