Article • Imaging tumour metabolism
Hyperpolarised MRI boosts cancer diagnosis
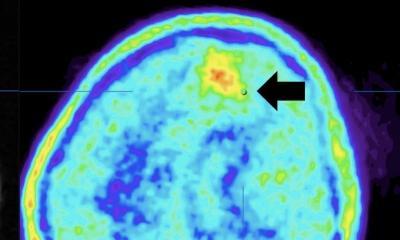
Tumour metabolism can be imaged with MRI as a technique to help determine cancer aggressiveness and response to therapy. The work by a UK-based group, on probing cancer metabolism non-invasively with clinical hyperpolarised carbon-13 MRI, can detect metabolic changes in the tumour. As metabolic changes occur much earlier than change in tumour size, this could have implications for quicker assessment of tumour response to treatment.
Report: Mark Nicholls

While still at an explorative stage, their advances on the use of MRI to image tumour metabolism were outlined at the recent Garmisch symposium of MR innovation by group leader Professor Ferdia Gallagher, who is Professor of Translational Imaging and Honorary Consultant Radiologist in the Department of Radiology at the University of Cambridge and Addenbrooke’s Hospital. He explained that cancer metabolism differs from normal tissue in that most cancers take up large quantities of glucose and convert it into the molecule lactate.
While that phenomenon can be detected using FDG-PET (fluorodeoxyglucose), his group is exploiting this mechanism using hyperpolarised MRI. This is an emerging method to non-invasively assess tissue metabolism using pyruvate – a breakdown product of glucose - as a probe to visualise tumour lactate formation. To achieve this, pyruvate is labelled with the non-radioactive isotope carbon-13 (13C) and lowered to a temperature close to absolute zero. Under these extreme conditions, the pyruvate and 13C become hyperpolarised meaning the signal to noise ratio detectable with MRI is significantly increased by several orders of magnitude. Following this, the pyruvate is warmed to body temperature and injected into the patient. “We can image the pyruvate reaching the tumour and the lactate forming in real time over the timescale of the experiment - which lasts a few minutes,” said Professor Gallagher. “We have shown recently that more aggressive breast cancer produces higher lactate levels and that when you treat that breast cancer, there is a reduction in this lactate. We are also interested in the brain where we can detect two forms of metabolism simultaneously: firstly, lactate as a consequence or reductive metabolism, and we also see bicarbonate forming as a result of oxidative metabolism. That potentially opens up the possibility that we can monitor metabolic changes in brain disease, as well as informing on treatment.”
However, there are challenges to what is a highly-technical process. To conduct the research, the Cambridge team created the first hyperpolarised 13C pharmacy in Europe. The process also requires a dedicated clean room space and radiopharmacy, and staff have to be trained to a high technical standard to prepare and deliver the 13C labelled pyruvate in a completely sterile environment. “In addition,” he added, “there are regulatory considerations and the challenge of identifying the best applications where the technology will make the biggest impact. But a counterpoint is that we are able to assess metabolism in a way that we have never been able to do so before.”
While FDG-PET allows visualisation of that in the tissue with greater sensitivity, the advantage with MRI is that it is able to detect individual metabolites and also measure metabolites forming dynamically in real time. “In the brain, we see the pyruvate perfusing in, and lactate and bicarbonate forming,” he said. “That is not possible with PET.” Imaging tumour metabolism with MRI is already showing potential advantages, particularly as it is known that metabolic changes occur much earlier than changes in tumour size.
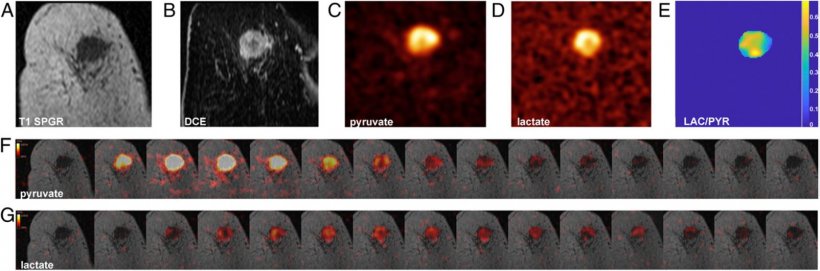
Image source: F Gallagher et al., PNAS 2020 (CC BY-NC-ND 4.0)
Tumour size changes can be quantified by CT scans repeated over weeks or months, but the Cambridge group has evidence from animal models that the metabolic changes occur within hours or days. This has implications for quicker tumour assessment to rapidly adapt treatments.
Imaging tumour metabolism with MRI for non-invasive assessment of tumour aggressiveness also has advantages over invasive biopsies. While biopsy tissue can be examined in great detail, it does not allow the oncologist to look at the whole tumour. Professor Gallagher said: “A feature of cancer is the more aggressive tumours tend to be more heterogenous and these aggressive tumours are often less likely to respond to therapy. Imaging metabolism allows us to look at heterogeneity on a metabolic level.”
The group has evidence from prostate and breast cancer that more aggressive tumours can be seen by examining metabolism in this way, and thus deliver the opportunity to stratify them based on metabolism. Evidence is building on the ability to see treatment response using this approach. A big advantage of imaging tumour metabolism with MRI is that it does not involve radiation. This will be of benefit when imaging children, women of reproductive age, or in patients with tumours where multiple repeat studies are required.
*The group’s work was recognised with the MRI Award at the Garmisch meeting, which Professor Gallagher accepted in recognition of all who had contributed to the project.
Profile:
Professor Ferdia Gallagher is Professor of Translational Imaging and a Cancer Research UK Senior Cancer Research Fellow and Honorary Consultant Radiologist in the Department of Radiology at the University of Cambridge and Addenbrooke’s Hospital. With a speciality in Oncological and Molecular Imaging, MRI, PET, he is an academic radiologist and leads the Clinical Molecular Imaging Group in the Department of Radiology. His laboratory develops new functional and molecular imaging methods to detect cancer and early response to therapy and is especially interested in methods to probe cancer metabolism non-invasively.
05.03.2021