Sponsored • Chronic liver disease
A looming pandemic and the need for physician partnerships to leverage non-invasive testing
By Dr. Katherine Soreng, Global Director of Clinical Education, Siemens Healthineers
Even as we battle one pandemic (Covid-19), we sit on the cusp of another. Europe has one of the highest burdens of chronic liver disease (CLD) in the world, driven largely by alcohol overconsumption, viral hepatitis, and obesity.1 Furthermore, non-alcoholic fatty liver disease (NAFLD) is increasingly common and is a significant contributor to CLD – especially in people with diabetes, where its prevalence is 68 %.2 Moreover, the number of adults with diabetes continues to climb.

NAFLD encompasses a spectrum of disease – from simple steatosis (fat buildup in the liver) to inflammatory damage associated with non-alcoholic steatohepatitis (NASH), fibrosis, cirrhosis, and liver-related events (LREs). NASH develops in about 20 % of people with NAFLD and elevates risk, but data clearly implicates advancing fibrosis – which can occur in both NAFLD and NASH – as the primary predictor of LREs. Assessing at-risk patients for progressive fibrosis, therefore, is essential if we are to improve earlier management and outcomes. “We are now seeing the burden from chronic liver disease that really started decades ago,” explains Prof. Manuel Romero-Gómez of Virgen del Rocio University Hospital in Seville. “NAFLD-related chronic liver disease is becoming pandemic – and without implementing measures for appropriate referrals, the patient load could quickly become unmanageable.”
Earlier recognition is key
CLD typically evolves over many years and often is clinically silent until there is an acute deterioration in liver function. According to Prof. Romero-Gómez, “We must increase identification of those patients with advancing fibrosis before they present with an LRE because that is when intervention measures are more effective.” In short, diagnoses tend to occur late, when only limited therapeutic options are available.3 Interceding before acute deterioration may slow – and even reverse – disease progression.
Without a substantial focus on earlier detection in at-risk populations, the coming decade will likely produce a colossal burden of advanced NAFLD-associated CLD. It is imperative that physicians assess their patients for NAFLD and advancing disease – especially in the primary care setting, where timely referral to hepatologists and other appropriate specialists may improve outcomes.
Fibrosis drives disease progression
That is where NITs can transform patient management – by supporting identification of that subset of patients truly in need of referral
Manuel Romero-Gómez
Liver fibrosis is active tissue scarring caused by the deposition of extracellular matrix proteins, including collagen, and occurs in most types of CLD. Although repair mechanisms within the liver can drive significant reversal of damage, these mechanisms also can become overwhelmed by advancing fibrosis.
Biopsy has been used for decades to detect liver fibrosis but has drawbacks – including invasiveness, limited testing volume, and the potential for flaws in diagnostic accuracy due to sampling issues and variation in observer interpretation. Non-invasive testing (NIT) that uses biomarkers, or imaging, as an alternative diagnostic tool for fibrosis assessment holds the potential for earlier detection – and for dramatically transforming CLD management – but it requires close coordination between physicians and specialties.
NITs that use blood-based biomarkers offer the greatest capacity for routinely assessing large numbers of patients for liver fibrosis. Easy-to-use diagnostic pathways that employ these types of NITs have been identified – and data shows that using them leads to a substantial increase in the detection of advanced fibrosis or cirrhosis, while also delivering a marked decrease in inappropriate referrals.4 Prof. Romero-Gómez explains why this is important: “Keep in mind that the average primary care physician is seeing 100 patients to detect two or three at high risk of advanced fibrosis. That is where NITs can transform patient management – by supporting identification of that subset of patients truly in need of referral.”

Biomarkers fall into two categories – indirect markers that assess for evidence of damage or inflammation, such as FIB-4, and direct markers, which detect for evidence of active fibrosis.
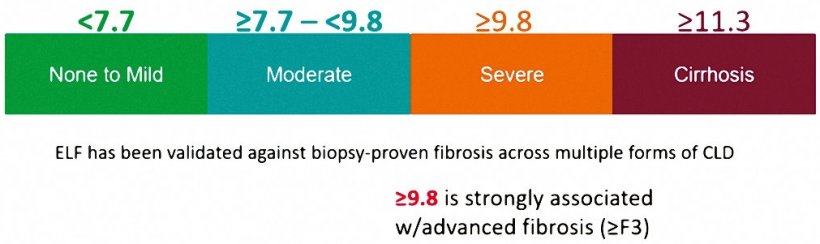
- The best-studied of the direct biomarkers is the ELF Test – a fully automated algorithm-based blood test that assesses for three direct markers of active fibrosis. → Table 1.
- Notably, it requires only a single routine serum sample and produces a simple-to-interpret numeric score. → Figure 1.
- More than 15 years of studies document ELF’s performance. NAFLD diagnostic pathways incorporating ELF have shown a substantial increase in detection as well as a decrease in inappropriate referrals. → Figure 2.
Unlike biopsy or imaging, which assess for existing damage, or indirect markers, which reflect damage or inflammation, ELF biomarkers are associated with active fibrosis, supporting both diagnostic and prognostic utilities. “NITs are providing valuable tools and associated guidelines for the detection of advanced fibrosis and cirrhosis,” says Prof. Romero-Gómez. “A simple blood test like ELF, or ELF in conjunction with a test like FIB-4, offers the greatest facility for assessing the high volume of at-risk patients presenting in primary care or related settings. As a hepatologist, I can be very confident that at-risk patients coming to me with a high ELF score benefit from the referral, whereas those with lower ELF scores can remain effectively managed in their current clinical setting.”
A collaborative approach is imperative
All told, the clinical and economic burdens of NAFLD in Europe are particularly high and will likely increase as its incidence continues to rise. Earlier diagnosis and intervention hold the potential for reducing future healthcare – and human – costs. To be effective, routine testing for evidence of disease progression relies heavily on provider awareness and cross-specialty collaboration. “Since most of our referrals come from primary care or specialties like GI or diabetology, it is essential that we partner in a peer-to-peer fashion to optimize a diagnostic referral pathway,” emphasizes Prof. Romero-Gómez, who advocates for a close, coordinated partnership between hepatologists and those clinicians most likely to initially encounter patients at-risk for CLD. “NITs like ELF will be key to facilitating that partnership.”
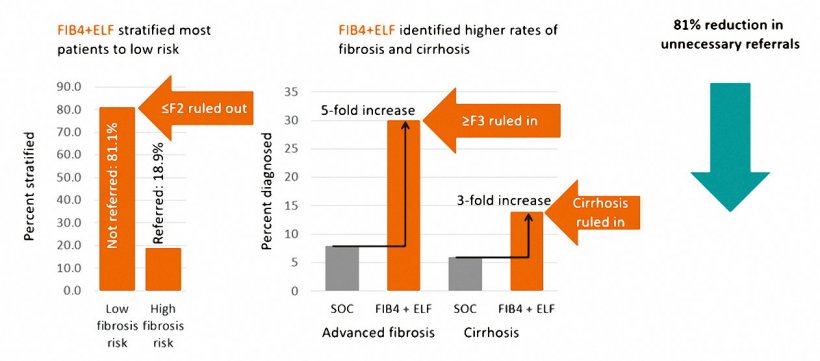
Figure 2. Outcomes Summary4
References
- Pimpin L, Cortez-Pinto H, Negro F, et al. Burden of liver disease in Europe: Epidemiology and analysis of risk factors to identify prevention policies. Journal of Hepatology. 2018;69(3):718-735. doi:10.1016/j.jhep.2018.05.011
- Younossi ZM, Golabi P, de Avila L, et al. The Global Epidemiology of NAFLD and Nash in patients with type 2 diabetes: A systematic review and meta-analysis. Journal of Hepatology. 2019;71(4):793-801. doi:10.1016/j.jhep.2019.06.021
- Wong RJ, Kachru N, Martinez DJ, Moynihan M, Ozbay AB, Gordon SC. Real-world comorbidity burden, health care utilization, and costs of nonalcoholic steatohepatitis patients with advanced liver diseases. Journal of Clinical Gastroenterology. 2020; Publish Ahead of Print. doi:10.1097/mcg.0000000000001409
- Srivastava A, Gailer R, Tanwar S, Trembling P, Parkes J, Rodger A, Suri D, Thorburn D, Sennett K, Morgan S, Tsochatzis EA, Rosenberg W. Prospective evaluation of a primary care referral pathway for patients with non-alcoholic fatty liver disease. J Hepatol. 2019 Aug;71(2):371-378. doi: 10.1016/j.jhep.2019.03.033. Epub 2019 Apr 6. PMID: 30965069
09.12.2021