News • Electrostatic interactions
New research exposes weak spot in SARS-CoV-2
Northwestern University researchers have uncovered a new vulnerability in the novel coronavirus’ infamous spike protein — illuminating a relatively simple, potential treatment pathway.
The spike protein contains the virus’ binding site, which adheres to host cells and enables the virus to enter and infect the body. Using nanometer-level simulations, the researchers discovered a positively charged site (known as the polybasic cleavage site) located 10 nanometers from the actual binding site on the spike protein. The positively charged site allows strong bonding between the virus protein and the negatively charged human-cell receptors.
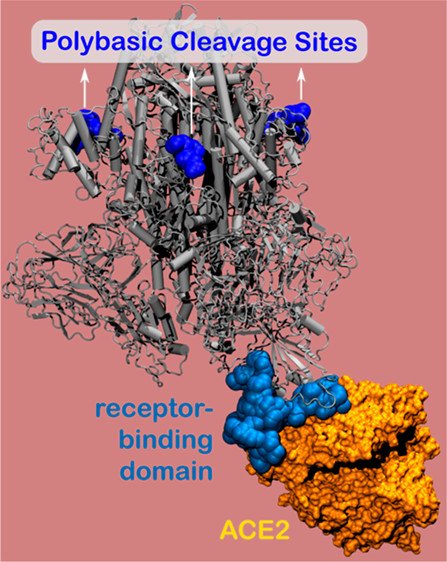
Image credit: Northwestern University
Leveraging this discovery, the researchers designed a negatively charged molecule to bind to the positively charged cleavage site. Blocking this site inhibits the virus from bonding to the host cell. “Our work indicates that blocking this cleavage site may act as a viable prophylactic treatment that decreases the virus’ ability to infect humans,” said Northwestern’s Monica Olvera de la Cruz, who led the work. “Our results explain experimental studies showing that mutations of the SARS-CoV-2 spike protein affected the virus transmissibility.” Olvera de la Cruz is the Lawyer Taylor Professor of Materials Science and Engineering in Northwestern’s McCormick School of Engineering. Baofu Qiao, a research assistant professor in Olvera de la Cruz’s research group, is the paper’s first author.
Made up of amino acids, SARS-CoV-2’s polybasic cleavage sites have remained elusive since the COVID-19 outbreak began. But previous research indicates that these mysterious sites are essential for virulence and transmission. Olvera de la Cruz and Qiao discovered that polybasic cleavage site is located 10 nanometers from human cell receptors — a finding that provided unexpected insight. “We didn’t expect to see electrostatic interactions at 10 nanometers,” Qiao said. “In physiological conditions, all electrostatic interactions no longer occur at distances longer than 1 nanometer.”
“The function of the polybasic cleavage site has remained elusive,” Olvera de la Cruz said. “However, it appears to be cleaved by an enzyme (furin) that is abundant in lungs, which suggests the cleavage site is crucial for virus entry into human cells.” With this new information, Olvera de la Cruz and Qiao next plan to work with Northwestern chemists and pharmacologists to design a new drug that could bind to the spike protein.
Source: Northwestern University
18.08.2020