Article • Multi-drug resistance
Aiming for anti-biofilm strategies
The association of biofilms with multi-drug resistance has highlighted the need for further understanding of biofilms and for anti-biofilm strategies.
This September, in a satellite symposium sponsored by Heraeus Medical, manufacturer of products for orthopaedic surgery and traumatology, at the 35th Annual meeting of the European Bone and Joint Infection Society, in Oxford, the latest knowledge of biofilms and local methods of overcoming the challenge of infection was discussed.
Biofilm-related implant malfunction
In orthopaedics, bacteria may colonise a surface for months or years but remain undetected until they trigger an immune response and signs of infection. During this time, there may be low-grade infection, and symptoms such as persistent pain, stiffness, lack of range-of-movement, fibrosis, ‘aseptic’ implant loosening and non-union of fractures.
Biofilms and multi-drug resistance
A mature biofilm can form within 24 hours of contact with an implant and can even penetrate the surface of titanium. As the biofilm matrix is more abundant than the bacteria themselves, the biofilm must be disrupted in order to expose the bacteria to antimicrobial agents.
Multi-drug resistance may also be related to biofilm formation, for example, methicillin-resistant Staphylococcus aureus (MRSA) produces more biofilm than methicillin-susceptible S. aureus (MSSA), and therefore biofilms may magnify the cost, suffering and mortality associated with antibiotic resistance.
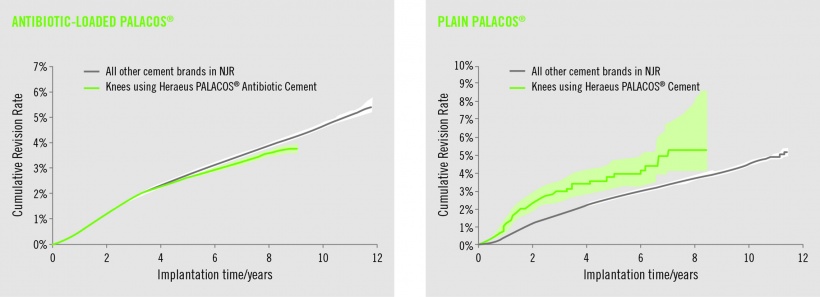
Superior outcome in hip and knee arthroplasty through antibiotic-loaded bone cements (ALAC)
In addition to the well-documented benefit of bone cements such as Palacos R+G in total hip replacement (THR), registry data was presented showing that significantly fewer revisions are required when that bone cement was used in total knee replacement surgery, compared to all other bone cements (Data on file. National Joint Registry (NJR, www.njrcentre.org.uk) data licensed to Heraeus Medical GmbH, Wehrheim). Palacos R+G with gentamicin also significantly outperforms plain Palacos.
Significant reduction of surgical site infections (SSIs) through dual antibiotic-loaded bone cements
It is particularly important to optimise strategies for reducing peri-prosthetic joint infections (PJIs) in high risk patients, but which bone cement best overcomes the current challenges in PJI prevention? A quasi-randomised, double blind study was carried out, comparing the two bone cements, Palacos R+G and Copal G+C, in patients requiring haemiarthroplasty for a fractured neck of femur. There was a significant reduction in both deep and superficial SSIs with Copal G+C compared with Palacos R+G (Tyas B, Marsh M, Molyneux C. et al. Abstract 59 presented at the Oxford meeting) explained by the previously-observed high antibiotic concentrations in wound fluid.
Extrapolating the reduction in infection rates to the entire UK hip haemiarthroplasty population would lead to healthcare savings of over £4 million.
Dual antibiotic-loaded cement does not affect antibiotic resistance profiles
A concern regarding the use of antibiotic loaded bone cements is whether they cause antibiotic resistance. The study reported no significant differences in resistance between Palacos R+G and Copal G+C. The only cases of resistance with Copal G+C were to gentamicin and clindamycin, which is as expected.
Resistance to other standard antibiotics remained low. Furthermore, Copal G+C completely eradicated infection caused by Corynebacterium species and S. aureus.
Source: Heraeus Medical manufacturer of medical products for orthopaedic surgery and traumatology. The products list includes bone cements.
25.07.2017





