Perfusion imaging: The future of CT?
CT scanners now nicely cover morphology. The challenge is moving to CT functional imaging without frying patients

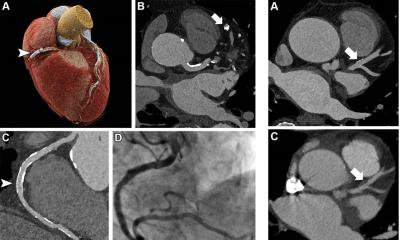
Cardiac scanning was the driving force behind recent developments in computed tomography (CT) that saw the introduction of multi-detector imaging as well as innovations such as dual-source flash scanners and wide 320-slice detectors. The next five years will see the development of a combination of CT angiography (CTA) and CT perfusion as a onestop shop for cardiologists, according to Professor Mathias Prokop. Radiologists will be able to offer examinations including stress tests capable of predicting the presence of ischaemia equally well as SPECT. Prof. Prokop and his group at the Radboud University Medical Centre are looking further to a day when CT perfusion can offer functional diagnosis throughout the body, starting with applications in the brain but spreading to oncologic and functional imaging.
The challenge is to create goodquality perfusion scans at an acceptable radiation dose. If perfusion imaging was done using conventional settings for each time point of a perfusion series, then radiation dose would have to be multiplied with the number of scans, and thus be larger than current techniques by a factor of 20-40. Since frying patients is no option, low-dose approaches are being developed that can achieve perfusion imaging at competitive dose, but these approaches so far struggle with reduced image quality. His group sees dose and image quality as the key issues and is working on developing techniques for highquality perfusion at acceptable dose levels. ‘Widespread use of perfusion imaging is not acceptable if we can’t bring down the dose,’ he said. ‘That’s the message.’
So, now that the Slice Wars have settled, where are we with CT imaging?
Prof Prokop: ‘Cardiac imaging used to be the driving force behind new CT developments. Today we can deliver pretty good quality imaging of cardiac morphology in almost all cases, though the technique is not completely fool proof. All the new generation scanners beyond 64-slice are capable of good-quality cardiac work. Cardiac imaging is especially simple with the scanner we use, which is a Toshiba 320-slice unit. In a fraction of patients there’s a bit of a problem left with motion correction. ‘GE Healthcare recently introduced a software approach to estimate the movement of the coronary arteries and use this information during image reconstruction to counteract this motion and improve sharpness and contour delineation of the coronaries. I expect this technology to spread to the other vendors who are already playing with it. Philips it was the first to publish, but hasn’t yet implemented the technique in their clinical software.
‘A big issue we have currently with cardiac CTA is that cardiologists tell us it’s nice to know the coronary morphology, but that it is more important to know whether a stenosis is functionally relevant. This information is important for deciding whether to treat this stenosis or not. Until very recently we were not very good at answering this question because, while we could see there was a stenosis, we could not say anything about collateral vessels or whether the stenosis caused a malperfusion of the myocardium.’
Is CT really a viable modality to answer this question?
‘Assessing the functional significance of a stenosis is a topic that is still under development and will remain so for the next few years, but it will get done. The current technique uses adenosine stress testing and a scan at a time point where there is the biggest difference in perfusion between normal and ischaemic myocardium. The tech nique has been studied, for example, at John’s Hopkins and been tried out in the Core-320 multicentre trial. CT perfusion turns out to predict ischaemia similarly well as SPECT CT, but further optimisation seems warranted. In principle, the technique can be used on any modern scanner but we find it easy on our 320-slice unit because of its ability to perform snapshot imaging even at the high heart rates encountered at stress testing. ‘Once the technique has matured, CT will act as a one-stop shop for patients with suspected ischaemia who now undergo a wide variety of tests ranging from bicycle stress testing to ultrasound, SPECT, MR or catheter coronary angiography. The CT workup would start with CTA to see if there are abnormalities. If there is a potentially significant stenosis, then a CT perfusion stress test will show if the stenosis is relevant and needs to be treated.’
In recent investigations you have taken CT perfusion beyond the heart. What potential do you see?
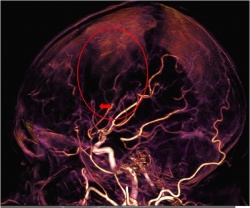
‘What’s coming next is dynamic perfusion imaging that consists of a series of short scans covering the target area. It can be done with most scanners with decent results, though again there is an advantage in covering an entire target, such as the brain or liver, with a wide detector that gives good signal-to-noise at a relatively low dose. ‘These perfusion scans have been successfully used in stroke imaging to discriminate between infarct core and penumbra and to identify proximal vessel occlusions that would warrant intra-arterial stroke treatment. We see increasing indications in oncologic imaging: we’d like to be able to distinguish early between responders and non-responder to biologicals, the expensive but powerful new class of oncologic drugs.
‘This would allow for substantial savings if treatment is stopped early and non-responders are put on alternative treatment. This appears feasible because CT perfusion is able to see an early drop in tumour perfusion in responders. Other applications of CT perfusion will include treatment planning for head and neck tumours, lungs, kidneys and tumour differentiation.'
‘However, the crucial factor for success is to keep radiation dose at bay while providing sufficient image quality. What we are working on now is to make sure that CT perfusion can be done without over-dosing the patient. Currently a CT perfusion of the brain scan will cause an effective radiation dose in the range of 2 or 3 mSv, which does not sound too bad. However, the local dose, for example to the eye lens, is much higher – well above 100 mGy, which is substantial. ‘We are trying a couple of approaches, and while we are not there yet, results look promising. We are now acquiring such low dose images during the perfusion series that a single image is almost unusable for diagnosis. The challenge is to understand how we can retrieve relevant information from these ultra-low dose, lousy-looking images.
‘One approach, for example, is a 4-D noise filter that can improve the image quality quite dramatically by using not only 2-D or 3-D information but data from the whole acquisition series. We are now already able to look at all the dynamic information: inflow and outflow of contrast via arteries and veins, and areas with delay in perfusion as an indirect sign of obstruction or other abnormalities. ‘One of our team members, Ewoud Smit, has shown that a relatively simple technique that we call timinginvariant CTA is able to depict collaterals much better than conventional CTA. He has found that if you do not see collaterals on a timing-invariant CTA the outcome of a stroke patient is invariable bad. Since conventional CTA is a looking at a particular moment in time, collaterals may not yet be enhanced during the time of acquisition. Timing-invariant CTA, on the other hand, uses data from the whole 4-D series and will display vessels impendent of when contrast arrives. The result is that you cannot rely on a conventional CTA if it does not display collaterals – they might be still there but not yet enhanced, while the timing-invariant technique will always show them if they are there at all.’
You’ve worked in Japan with Toshiba, on phantom models relating to the ViSION Edition scanner. What’s the goal?
‘We have a Toshiba grant to develop a phantom that allows us to play around with the CT acquisition parameters without having to expose patients to radiation. It allows us to determine how to choose scanning and post-processing parameters to optimise the image quality. We needed models that allowed us to study the influence of each variation we are testing on the accuracy of perfusion measures, noise, spatial resolution, signal-to-noise ratios and, ultimately, visibility of small perfusion abnormalities, such as lacunar infarcts. ‘The phantoms need to be organ-specific to replicate the noise that comes from real scanning. The noise in the brain is, of course, different from the chest, for example. We now have a phantom for the brain. We will probably develop such a phantom for the abdomen, and then one for the chest. Toshiba has fully embraced this approach.’
You are updating your best-selling book ‘Spiral and Multi-slice Computed Tomography of the Body’.
What can we expect to see?
‘We hope it will come out at the end of this year. Our goal is to include the very latest information. What will be significant in the new edition is the latest technology, such as perfusion imaging, the new field for development, as well as the related subject of dual-energy and subtraction imaging for contrast-enhanced studies. We are now applying the same techniques we use to extract information from perfusion scans to subtraction imaging, another powerful tool for creating iodine maps at various time points after contrast injection. The resulting high-resolution maps look pretty cool and will probably revolutionise the way we look at contrastenhanced studies.’
PROFILE
Mathias Prokop trained as a radiologist at Hanover Medical School, Germany and gained a BSc in Physics at Marburg University, Germany. From 1998 he was an Associate Professor of Radiology at the University of Vienna Medical School. He went to the Netherlands in 2002 and became Professor of Radiology at UMC Utrecht in 2004. In 2009 he was appointed Professor of Radiology at Radboud University Nijmegen as well as Chairman of the Department of Radiology. Dr Prokop is an expert in body imaging with a special focus on multislice CT and new imaging technologies. Over the past decade he has concentrated on chest screening using CT (cancer, cardiovascular disease, COPD) and has been a major player in the Dutch-Belgian lung cancer screening trial (NELSON).
09.03.2013