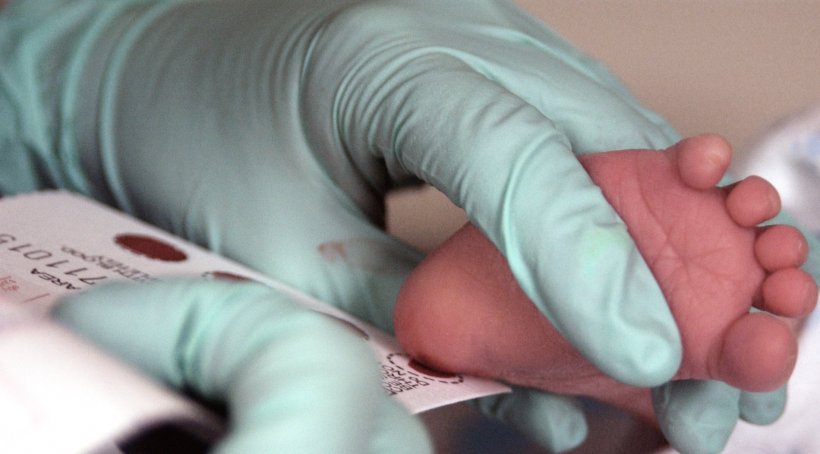
Image source: U.S. Air Force photo/Staff Sgt Eric T. Sheler, Phenylketonuria testing, marked as public domain, more details on Wikimedia Commons
Article • Mass spec, qPCR and more
Newborn screening: early detection – new diagnostic possibilities
Since its introduction around 60 years ago, the screening of newborn babies for immune, hormone and metabolic disorders has prevented many people from experiencing severe disease progression. The scope of systematic early testing has been significantly enhanced through mass spectrometry (MS). In our interview, Professor Uta Ceglarek, one of the driving forces behind the introduction of MS procedures in newborn screening, outlines new diagnostic possibilities offered by MS and qPCR procedures for screening – and also the ethical limits and possible dangers of new regulations for their implementation.

The foundations for using drops of blood from newborn babies to make predictions about the development and prevention of diseases later in life were laid in the 1960s with the systematic screening of babies for phenylketonuria (PKU). Professor Ceglarek deems this a success story, confirming, ‘It has allowed us to diagnose diseases before symptoms develop.’
The expert views the inherited metabolic disorder PKU as a prime example of the importance of screening: Those affected lack an important enzyme which results in the amino acid phenylalanine not being converted into tyrosine. When phenylalanine is ingested through food it accumulates in the body – with serious consequences. ‘Once these diseases take hold, the damage they cause is often irreversible.’
Due to the lack of tyrosine, untreated PKU can lead to severe impact on brain development, with resulting intellectual disabilities. Prior to the introduction of newborn screening, those affected required lifelong care and, in the most severe cases, had to be looked after in specialist care facilities. However, when PKU is detected early, the disorder can be managed well, with strict dietary measures, and specifically through the reduction of protein in the diet. ‘The first patients who had PKU diagnosed during newborn screening are now aged over 40 and, due to early detection of the disease, lead normal lives,’ Ceglarek points out.
MS/MS covers the lion’s share
The range of diseases diagnosed through screening has increased significantly through the use of mass-spectrometry, and specifically through the tandem procedure MS/MS. The so-called enhanced newborn screening (funded by the statutory health insurers in Germany since 2005), currently includes 19 inherited diseases, nine of which are diagnosed via MS/MS [source: https://www.screening-dgns.de/richtlinien.php]. Additionally, PCR diagnosis, a procedure which has become more widely known to the public during the Corona pandemic, is used to diagnose severe combined immunodeficiency and spinal muscular atrophy.
The prerequisite for screening inclusion is the existence of a reliable diagnostic procedure and the respective disorder being treatable. Although curative care is not possible in all cases, such as in the case of mucoviscidosis, early detection helps to delay the progression of the disease and to increase the quality of life for those affected.
In some cases, the therapeutic opportunities improve over the course of time, meaning that screening of newborns still makes sense. Currently, this includes spinal muscular atrophy (SMA), which can be safely diagnosed via PCR, but which cannot be cured. Zolgensma (AVXS-101), a new medication introduced only this year, has brought a cure for muscular atrophy within tangible reach. ‘Newborn screening for SMA started on 1 October 2021,’ Ceglarek reports. ‘The screening community is now observing with great interest how successful pre-symptomatic gene therapy will be.’
New legislation threatens diagnostic options
The new version of the EU In Vitro Diagnostic Medical Device Regulation (IVDR), which will finally come into force in 2022, will make the use of new, but also of already established, self-developed procedures much more difficult, Ceglarek says.
If the validation of these tests is this comprehensive, it will no longer be viable to offer them, so important diagnostic procedures will no longer be commercially available and cannot be offered as LDTs
Uta Ceglarek
This also affects mass spectrometry. On the one hand, all procedures developed in the laboratory must be newly revalidated before they can be used on patients. ‘This means large, additional overheads for all parameters which need to be reviewed,’ Ceglarek points out. On the other hand, the regulation prescribes the use of CE marked, commercially available laboratory tests. Even self-developed, ‘in-house’ solutions, so-called LDTs, can only be used when there is no equivalent product available in the market, and only if they can prove full CE conformity. ‘The IVDR is not about economic efficiency, meaning that the use of a different procedure must always be justified with reasons other than cost efficiency. This means that in case of comparable analytical characteristics, the commercial method must always be used.
This also has devastating effects on molecular diagnostics, which, to a large part, works with self-developed reagents: ‘In our laboratory, we are currently having to evaluate which LDTs we can realistically still offer,’ Ceglarek explains, adding: ‘If adherence to the IVDR had been compulsory at the start of the Corona pandemic, the initial supply of Covid LDTs would have taken a lot longer, because each procedure would have needed extensive validation before being used on patients.’
The new regulation could therefore turn into a nail in the coffin for many laboratory-diagnostic procedures, such as those for diagnosis of rare diseases: ‘If the validation of these tests is this comprehensive, it will no longer be viable to offer them, so important diagnostic procedures will no longer be commercially available and cannot be offered as LDTs,’ Ceglarek says, appealing to those in charge, and calling for the IVDR to be modified before the end of the transition period.
Profile:
Professor Uta Ceglarek is Deputy Director of the Institute of Laboratory Medicine, Clinical Chemistry and Molecular Diagnostics (ILM) at the University Hospital Leipzig, where she has headed the Newborn Screening Laboratory since 2005. Since 2000 she has worked on the use of mass-spectrometry diagnostics in newborn screening, for therapeutic drug monitoring and for metabolic indications. Ceglarek is also president of the German Society for Newborn Screening (DGNS) and Representative of the Section for Clinical Mass Spectrometry at the German Society for Clinical Chemistry and Laboratory Medicine (DGKL).
02.12.2021