Image source: Institute for Bioengineering of Catalonia (IBEC)
News • Urea-powered machines
Nanorobots to reduce bladder tumours by 90%
Bladder cancer has one of the highest incidence rates in the world and ranks as the fourth most common tumour in men.
Despite its relatively low mortality rate, nearly half of bladder tumours resurface within five years, requiring ongoing patient monitoring. Frequent hospital visits and the need for repeat treatments contribute to making this type of cancer one of the most expensive to cure. While current treatments involving direct drug administration into the bladder show good survival rates, their therapeutic efficacy remains low. A promising alternative involves the use of nanoparticles capable of delivering therapeutic agents directly to the tumour. In particular, nanorobots—nanoparticles endowed with the ability to self-propel within the body—are noteworthy.
Now, a study published in the journal Nature Nanotechnology reveals how a research team successfully reduced the size of bladder tumours in mice by 90% through a single dose of urea-powered nanorobots.
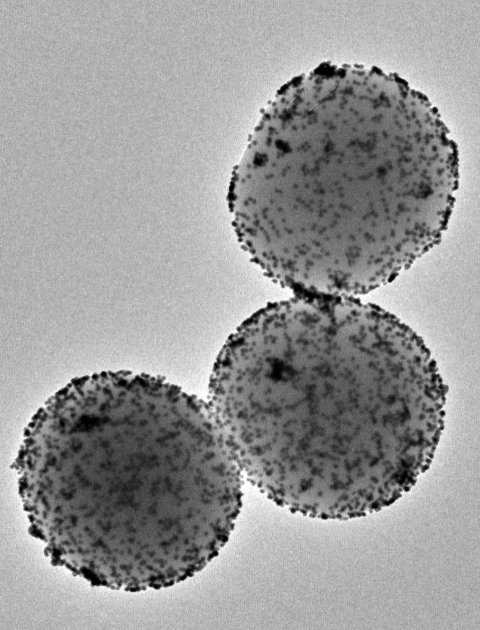
Image source: Institute for Bioengineering of Catalonia (IBEC)
These tiny nanomachines consist of a porous sphere made of silica. Their surfaces carry various components with specific functions. Among them is the enzyme urease, a protein that reacts with urea found in urine, enabling the nanoparticle to propel itself. Another crucial component is radioactive iodine, a radioisotope commonly used for the localized treatment of tumours.
The research, led by the Institute for Bioengineering of Catalonia (IBEC) and CIC biomaGUNE in collaboration with the Institute for Research in Biomedicine (IRB Barcelona) and the Autonomous University of Barcelona (UAB), paves the way for innovative bladder cancer treatments. These advancements aim to reduce the length of hospitalization, thereby implying lower costs and enhanced comfort for patients.
“With a single dose, we observed a 90% decrease in tumour volume. This is significantly more efficient given that that patients with this type of tumour typically have 6 to 14 hospital appointments with current treatments. Such a treatment approach would enhance efficiency, reducing the length of hospitalization and treatment costs,” explains Samuel Sánchez, ICREA research professor at IBEC and leader of the study.
The next step, which is already underway, is to determine whether these tumours recur after treatment.
In previous research, the scientists confirmed that the self-propulsion capacity of nanorobots allowed them to reach all bladder walls. This feature is advantageous compared to the current procedure where, after administering treatment directly into the bladder, the patient must change position every half hour to ensure that the drug reaches all the walls.
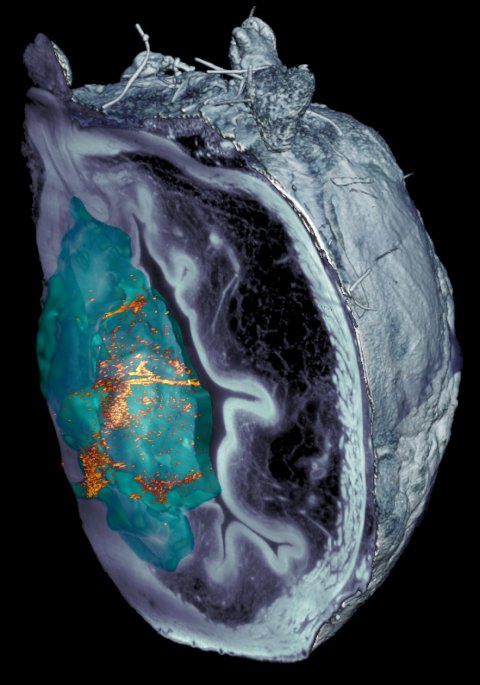
Image source: Institute for Bioengineering of Catalonia (IBEC)
This new study goes further by demonstrating not only the mobility of nanoparticles in the bladder but also their specific accumulation in the tumour. This achievement was made possible by various techniques, including medical positron emission tomography (PET) imaging of the mice, as well as microscopy images of the tissues removed after completion of the study. The latter were captured using a fluorescence microscopy system developed specifically for this project at IRB Barcelona. The system scans the different layers of the bladder and provides a 3D reconstruction, thereby enabling observation of the entire organ.
“The innovative optical system that we have developed enabled us to eliminate the light reflected by the tumour itself, allowing us to identify and locate nanoparticles throughout the organ without prior labelling, at an unprecedented resolution. We observed that the nanorobots not only reached the tumour but also entered it, thereby enhancing the action of the radiopharmaceutical,” explains Julien Colombelli, leader of the Advanced Digital Microscopy platform at IRB Barcelona.
Deciphering why nanorobots can enter the tumour posed a challenge. Nanorobots lack specific antibodies to recognise the tumour, and tumour tissue is typically stiffer than healthy tissue. “However, we observed that these nanorobots can break down the extracellular matrix of the tumour by locally increasing the pH through a self-propelling chemical reaction. This phenomenon favoured greater tumour penetration and was beneficial in achieving preferential accumulation in the tumour,” explains Meritxell Serra Casablancas, co-first author of the study and IBEC researcher. Thus, the scientists concluded that the nanorobots collide with the urothelium as if it were a wall, but in the tumour, which is spongier, they penetrate the tumour and accumulate inside. A key factor is the mobility of the nanobots, which increases the likelihood of reaching the tumour.
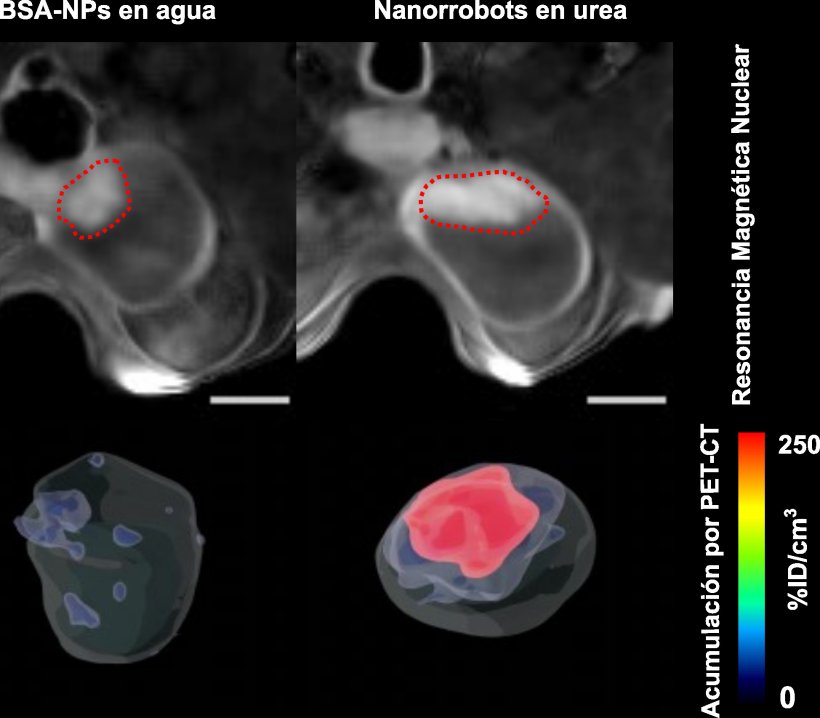
Image credit: CIC biomaGUNE
In addition, according to Jordi Llop, a researcher at CIC biomaGUNE and co-leader of the study, “The localized administration of the nanorobots carrying the radioisotope reduces the probability of generating adverse effects, and the high accumulation in the tumour tissue favours the radiotherapeutic effect.”
“The results of this study open the door to the use of other radioisotopes with a greater capacity to induce therapeutic effects but whose use is restricted when administered systemically,” adds Cristina Simó, co-first author of the study.
The study consolidates the results of over three years of collaborative efforts between various institutions. Part of the data stems from the doctoral theses of Meritxell Serra and Ana Hortelao, both researchers in IBEC’s Smart nano-bio-devices group, led by Sánchez. It also includes the thesis of Cristina Simó, co-first author of the study, who conducted her predoctoral research in the Radiochemistry and Nuclear Imaging Lab led by Jordi Llop at CIC biomaGUNE. The expertise of Esther Julián´s group at the UAB in the animal model of the disease is an additional contribution. Moreover, the project has received funding from the European Research Council (ERC) and the” la Caixa” Foundation.
The technology underlying these nanorobots, which Samuel Sánchez and his team have been developing for over seven years, has recently been patented and serves as the foundation for Nanobots Therapeutics, a spin-off of IBEC and ICREA established in January 2023. The company, founded by Sánchez, acts as a bridge between research and clinical application: “Securing robust funding for the spin-off is crucial to continue advancing this technology and, if all goes well, bring it to market and society. In June, just 5 months after the creation of Nanobots Tx, we successfully closed the first round of funding, and we are enthusiastic about the future,” highlights Sanchez.
Working with nanorobots has posed a significant scientific challenge in bioimaging techniques for visualizing these elements in tissues and the tumour itself. Common non-invasive clinical techniques, such as PET, lack the necessary resolution to locate these very small particles at a microscopic level. Therefore, the Scientific Microscopy Platform at IRB Barcelona employed a microscopy technique using a sheet of laser light to illuminate samples, allowing the acquisition of 3D images through light scattering upon interaction with tissues and particles. Upon observation that the tumour itself scattered part of the light, generating interference, the scientists developed a new technique based on polarized light that cancels out all scattering from the tumour tissue and cells. This innovation enables the visualization and location of nanorobots without the need for prior tagging with molecular techniques.
Source: Institute for Bioengineering of Catalonia
16.01.2024