Molecular Imaging for Alzheimer’s Disease May Be Available in Hospitals Within One Year
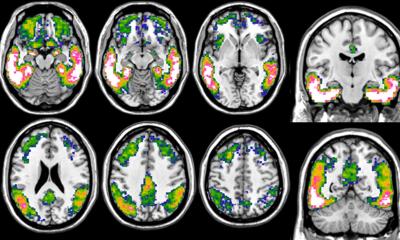
Researchers the world over are advancing positron emission tomography (PET) as an effective method of early detection for Alzheimer’s disease, a currently incurable and deadly neurological disorder. Three studies presented at SNM’s 58th Annual Meeting are providing new insights into the development of Alzheimer’s disease while opening the door to future clinical screening and treatments.

According to the World Health Organization, an estimated 18 million people worldwide are currently living with Alzheimer’s disease—a number projected to almost double by 2025.
“The aging population around the world is escalating exponentially. From a macro perspective, amyloid imaging with PET scans can help to ascertain the likelihood that individuals will deteriorate cognitively within a few years, thereby enabling more efficient channeling of health care resources,” says Kevin Ong, MD, lead author of a presented study and a research scientist at Austin Hospital, Melbourne, Australia. “From a micro perspective, planning and lifestyle modifications are possible for individuals who seek screening for Alzheimer’s disease.”
Molecular imaging of Alzheimer’s disease is focused on detecting and analyzing the formation of a naturally occurring protein in the brain called beta-amyloid, which researchers now say is directly involved in the pathology of Alzheimer’s.
“It turns out that increased amyloid is bad for cognition even in the healthy elderly,” says Michael Devous, Sr., PhD, director of neuroimaging for the Alzheimer’s Disease Center at UT Southwestern Medical Center, Dallas, Texas. “If you look at working memory, processing speed or fluent reasoning, three critical general domains of cognition, the more amyloid you have the worse your performance, and that’s after correcting for age.”
Not only is this important for imaging the disease, but it may also prove to be the key to amyloid-associated therapies and vaccines. Investigators warn that the beginning stages of the disease can precede symptoms of dementia as much as a decade or more. Imaging patients when they first show signs of mild cognitive impairment could be essential to determining their risk of future disease.
“For individuals who have already developed a measurable memory decline, a positive scan for amyloid is the most accurate predictor of progression to Alzheimer’s disease,” said Christopher Rowe, MD, a lead investigator for the Australian Imaging, Biomarkers and Lifestyle (AIBL) study of aging and professor of nuclear medicine at Austin Hospital, Victoria, Australia. “Amyloid imaging with PET scans is expected to be widely available soon for clinical practice. It will be an important new tool in the assessment of cognitive decline.”
Beta-amyloid accumulates as neurotoxic plaque in individuals preceding the onset of Alzheimer’s disease, and as a result, extensive neural tissues that control major mental and physical functioning—including memory, language and motor function—are decimated. In time the disease becomes debilitating and has a devastating impact on individuals’ quality of life.
Three ongoing studies presented at this year’s meeting involve several years of research based on hundreds of participants ranging widely in age, cognitive ability and stage of disease. Results of these studies show that amyloid plaques build up very slowly, by an estimated two to three percent per year, and that they are often already present in healthy older individuals—12 percent of those in their 60s, 30 percent of those in their 70s and 55 percent in those over the age of 80. In one study, about 25 percent of subjects over the age of 60 had amyloid plaques. The presence of significant amyloid buildup is linked to quicker memory decline and brain atrophy.
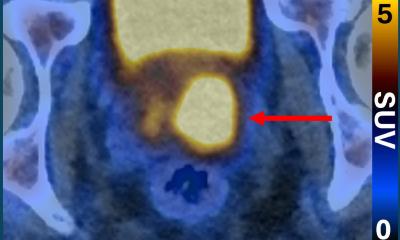
One of the major questions in Alzheimer’s imaging has been which imaging agent is best for amyloid plaque screening. Several studies have been conducted using 11C Pittsburgh Compound-B (11C-PIB), a PET imaging agent that binds to beta-amyloid in neural tissues, but two of the current studies are gauging the benefit of using F-18 labeled tracers (F-18 Florbetaben and F-18 Florbetapir), designed for routine clinical use. Both F-18 Florbetaben and F-18 Florbetapir are proving to be good predictors of progression to Alzheimer’s disease, and F-18 amyloid imaging agents are the likeliest agents to move forward into clinical practice in the near future.
Researchers estimate that amyloid imaging agents will be available for clinical use in fewer than 12 months. These and further studies will continue to amass data about the development of Alzheimer’s disease, and potential treatments could eventually stop and perhaps even prevent or reverse damage in the brain caused by Alzheimer’s disease.
Scientific Paper 119: K. Rodrigue, K. Kennedy, M. Devous, D. Park, UT Southwestern Medical Center, Dallas, TX; “Beta-amyloid in healthy aging: Regional distribution and cognitive consequences,” SNM’s 58th Annual Meeting, June 4–8, 2010, San Antonio, TX.
Scientific Paper 121: C. Rowe, K. Ellis, B. Brown, P. Bourgeat, R. Head, R. Martins, O. Salvado, C. Masters, D. Ames, V. Villemagne, Austin Hospital, Melbourne, Australia; University of Melbourne, Melbourne, Australia; Edith Cowan University, Perth, Australia; Australian e-Health Research Centre, CSIRO, Brisbane, Australia; National Ageing Research Institute, Melbourne, Australia; Mental Health Research Institute, Melbourne, Australia; “The consequences of Aβ deposition in ageing and Alzheimer’s disease: Results from 366 elderly subjects,” SNM’s 58th Annual Meeting, June 4–8, 2010, San Antonio, TX.
Scientific Paper 170: K. Ong, V. Villemagne, A. Bahar-Fuchs, F. Lamb, G. Chételat, C. Reininger, B. Putz, B. Rohde, C. Masters, C. Rowe. Austin Hospital, Melbourne, Australia; Bayer Schering Pharma, Berlin, Germany; Mental Health Research Institute, Melbourne, Australia; “Conversion from mild cognitive impairment to Alzheimer’s disease over 12 months: Predictive value of Aβ imaging with 18F-Florbetaben,” SNM’s 58th Annual Meeting, June 4–8, 2010, San Antonio, TX.
14.06.2011