Here - and the far horizons
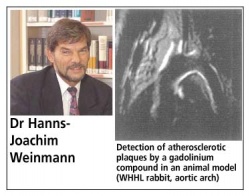
Dr Hanns-Joachim Weinmann, Diagnostics and Radiopharmaceuticals, Magnetic Resonance Imaging & X-Ray Research, Schering AG, examines what is feasible and what still lies only at a tantalising distance.
What is molecular imaging? Is it a buzzword to stimulate the imaginations of researchers, managers and analysts, or something real - a part of a daily routine in imaging departments? Honestly, nobody really knows. If we understand molecular imaging as the in vivo characterisation of biological processes at cellular and molecular levels, we are already fully in the future.
This article mainly reflects Schering’s view on the clinical imaging techniques (CT, MRI, and US). It is not an evaluation of SPECT, PET and NIR in detail.
Today’s MRI is far more than just another imaging modality; it offers not just morphological or anatomical details, it also gives physicians detailed information on the physiological status of the area of interest. An area of long T2 relaxation time, what does it mean? Because of our accumulated knowledge in reading such images, it could be an inflammatory process. Why do we see brain metastases as bright as a star after dosing gadolinium? The exobiotic contrast agent clearly delineates the breakdown of the blood-brain barrier, or an area without a blood-brain barrier, for example, a malign tumour. Following the first marketing authorisation for an MRI contrast agent (Magnevist) additional new indications are being investigated. How does it work that a so-called unspecific contrast agent elicits a specific contrast enhancement? The delayed enhancement in the myocardium is a sensitive marker of viability - obviously not too fancy, is it? We are already seeing changes at the cellular level, but we are simply not used to accepting this.
Targeting a plasma membrane bound protein or receptor has become a reality after the introduction of Primovist and Multihance. The gadolinium chelates bind specifically to a transporter located on the sinusoidal and bile canalicular surfaces of hepatocytes. To this day, the binding kinetics and the window to functional studies are just a little open - and by no means divulged.
Another example of today’s molecular imaging in MRI is superparamagnetic particles (SPIO), for example, Resovist. Those tiny aggregates, a product from nanotechnology, stimulate mononuclear phagocytes (Kupffer cells), the few nanometer large aggregates will be engulfed. Mononuclear phagocytes include the monocytes (in the bloodstream) and lymphocytes (mostly resident in lymphatic tissues); that’s why SPIO accumulate in the liver and also in the lymph nodes: in-vivo targeting of very specific cells. Using these superparamagnetic iron aggregates in stem cell research is just at the beginning. Stem cell can be labelled by SPIO in-vitro. When injected into the blood stream, cell trafficking can be monitored by MRI. In-vivo labelling of other specific cells, benign or malign, will be the next step into molecular imaging.
Researchers at Schering AG demonstrate that Gd-labelled antibodies can be used as highly specific tumour-seeking agents for MRI. However, such magic bullets didn’t make it into real medicine. Not because they where not specific enough, the commercial aspect still dominates its practicability. MRI is not a very sensitive technique so far as contrast efficacy is concerned. Micromolecular concentrations of gadolinium ions cannot be achieved after dosing just a few milligrams; consequently, the amount of antibodies becomes too large. Biotechnology is quickly developing and single chain fragments or small polypeptide will pave the way for disease-specific MRI agents.
EPIX, a small Cambridge/USA based pharmaceutical company just finished the phase I study of such a peptide-based gadolinium chelate. Equipped with a couple of high-relaxivity gadolinium chelates, the agent targets fibrin. This new compound is designed to detect pulmonary embolism, deep venous thrombosis and blood clots in the coronary and carotid arteries, to name a few relevant indications. Recently, water-soluble gadolinium endohedral metallofullerenes have been synthesised. Such Gd-fullerenols, with one or even two Gd atoms, exhibit the highest relaxivity ever measured per paramagnetic atom. Are these the tools we are looking for to make MRI molecular imaging even more specific?
Cell tracking in the living organism, using magnetically labelled cells, is under active investigation, and this research will open new opportunities in molecular imaging and contrast-enhanced MRI.
Ultrasound (US) is the most common imaging technology in medicine. Phospholipid-encapsulated, liquid perfluorodichlorooctane is the basis of a new, targeted ultrasound agent. When these submillimetre-sized particles were combined with a fibrin-seeking antibody, they could identify blood clots. Several groups showed that microbubbles could be guided to intravascular receptors of inflammation, atherosclerosis, and angiogenesis. Antibodies were linked to the coating material of the US microparticle.
One of the latest preparations, which contains gas entrapped in cyanoacrylate particles, produces a strong contrast when administered intravenously. Highly effective imaging techniques, such as stimulated acoustic emission, require only small quantities of this agent to produce pathology-specific enhancements.
New developments in nanotechnology will further improve the efficacy and potential of target-specific imaging agents. However, at least for the next decade classical small water-soluble agents will be the gold standard for most daily studies in CT and MRI. Other more sensitive imaging techniques, such as SPECT, PET or newly emerging near infrared imaging modalities will reach different levels of molecular imaging within coming years.
07.08.2006