Image source: Banbury et al., Science Advances 2023 (CC BY 4.0)
News • Point-of-care neurodiagnosis
Eye-safe laser technology to diagnose traumatic brain injury
The technology is expected to be developed into a hand-held device for use in the critical ‘golden hour’ after traumatic brain injury
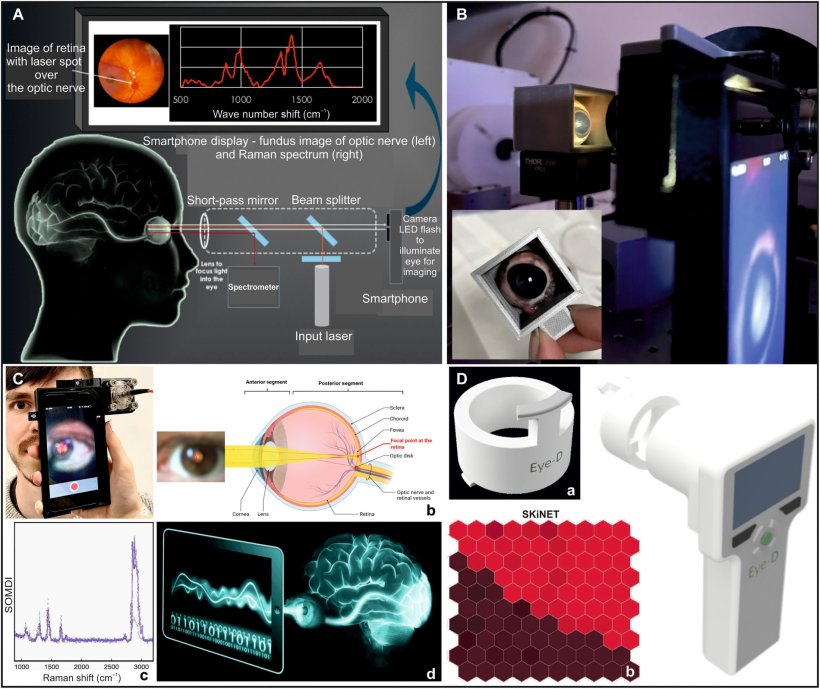
Described in Science Advances, the technique is dramatically different from other diagnostic methods and is expected to be developed into a hand-held device for use in the critical ‘golden hour’ after traumatic brain injury, when life critical decisions on treatment must be made. The device incorporates a class 1, CE marked, eye-safe laser and a unique Raman spectroscopy system, which uses light to reveal the biochemical and structural properties of molecules by detecting how they scatter light, to detect the presence and levels of known biomarkers for brain injury.
There is an urgent need for new technologies to improve the timeliness of TBI diagnosis, which is a leading cause of death worldwide. TBI is caused by sudden shock or impact to the head, which can cause mild to severe injury to the brain, and although it needs diagnosis and treatment as soon as possible to prevent further irreversible damage, it is hard to diagnose at the point of injury. Moreover, radiological investigations such as X-ray or MRI are very expensive and slow to show results.
"Early diagnosis of TBI is crucial, as life-critical decisions on treatment must be made with the first ‘golden hour’ after injury
Pola Goldberg Oppenheimer
Birmingham researchers, led by Professor Pola Goldberg Oppenheimer from the School of Chemical Engineering, designed and developed the novel diagnostic hand-held device to assess patients as soon as injury occurs. It is fast, precise and non-invasive for the patient, causing no additional discomfort, can provide information on the severity of the trauma, and will be suitable to be used on-site - at the roadside, on the battlefield or on the sports pitch - to assess TBI. "Early diagnosis of TBI is crucial, as life-critical decisions on treatment must be made with the first ‘golden hour’ after injury. However, current diagnostic procedure relies on observation by ambulance crews, and MRI or CT scans at a hospital – which may be some distance away," said Professor Goldberg Oppenheimer.
The device works by scanning the back of the eye where the optic nerve sits. Because the optic nerve is so closely linked to the brain, it carries the same biological information in the form of protein and lipid biomarkers. These biomarkers exist in a very tightly regulated balance, meaning even the slightest change may have serious effects on the ‘brain-health’. TBI causes these biomarkers to change, indicating that something is wrong.
Previous research has demonstrated the technology can accurately detect the changes in animal brain and eye tissues with different levels of brain injuries - picking up the slightest changes.1,2,3 The device detailed in the current paper detects and analyses the composition and balance of these biomarkers to create ‘molecular fingerprints’. The current study details the development, manufacture, and optimisation of a proof-of-concept prototype, and its use in reading biochemical fingerprints of brain injury on the optic nerve, to see whether it is a viable and effective approach for initial ‘on the scene’ diagnosis of TBI.
The researchers constructed a phantom eye to test its alignment and ability to focus on the back of the eye, used animal tissue to test whether it could discern between TBI and non-TBI states, and also developed decision support tools for the device, using AI, to rapidly classify TBIs.
The device is now ready for further evaluation including clinical feasibility and efficacy studies, and patient acceptability. The researchers expect the diagnostic device to be developed into a portable technology which is suitable for use in point-of-care conditions capable to rapidly determine whether TBI occurs as well as classify whether it is mild, moderate or severe, and therefore, direct triage appropriately and in timely manner.
References:
- Banbury, I. Styles, N. Eisenstein, E. R. Zanier, G. Vegliante, A. Belli, A. Logan, P. Goldberg Oppenheimer, Spectroscopic detection of traumatic brain injury severity and biochemistry from the retina. Biomed. Opt. Express 11, 6249–6261 (2020).
- Banbury, R. Mason, I. Styles, N. Eisenstein, M. Clancy, A. Belli, A. Logan, P. Goldberg Oppenheimer, Development of the self optimising kohonen index network (SKiNET) for Raman spectroscopy based detection of anatomical eye tissue. Sci. Rep. 9, 10812 (2019).
- Marro, A. Taubes, A. Abernathy, S. Balint, B. Moreno, B. Sanchez-Dalmau, E. H. Martínez-Lapiscina, I. Amat-Roldan, D. Petrov, P. Villoslada, Dynamic molecular monitoring of retina inflammation by in vivo Raman spectroscopy coupled with multivariate analysis. J. Biophotonics 7, 724–734 (2014).
Source: University of Birmingham
02.12.2023