Article • Predicting plaques
Exposing the secrets of the heart
Coronary interventions often rely more on art than science as the decision to treat a patient tends to be based on what clinicians can see, a subjective interpretation of cardiac imaging. Two new techniques have emerged for cardiovascular diagnostics that are enabling software to help surgeons and cardiologists measure, and thereby better manage cardiac disease. Both rely on powerful computer processing to expose the secrets of the heart.
Report: John Brosky
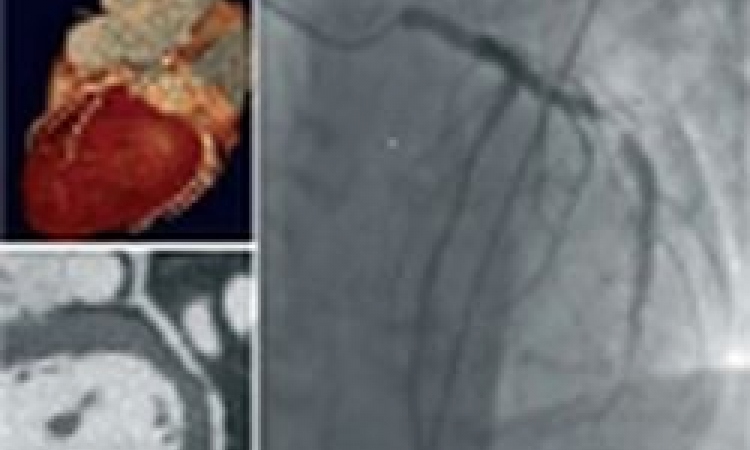
Bon-Kwon Koo MD, from the Seoul National University Hospital in South Korea, has successfully shown how building on technique recommended in cardiology guidelines, called fractional flow reserve (FFR), a retrospective computational analysis of CT exams can pinpoint the plaque rupture that, up to two years later, would cause a confirmed medical emergency for a patient.
The creator of the FRR technique, Nico Pijls MD, from the Catharina Hospital in Eindhoven, the Netherlands, has since developed a technique for the quantitative assessment of microcirculatory blood flow and resistance, the vital irrigation of heart muscle.
Without FFR, a cardiologist has to subjectively interpret fuzzy angiographic images to decide if the blockage is so severe it needs a stent to re-open the vessel. Introduced by Pijls 20 years ago, FFR is an invasive technique in which a catheter is pulled across a suspected coronary lesion to objectively measure differences in blood pressure on either side of a blockage and gives the cardiologist evidence to decide whether to place a stent or not.
Koo builds upon this critical measurement, but uses a non-invasive technique. Instead of pushing a catheter into the patient, a super-computer analysis of the patient’s CT angiography exam can determine blood pressure measures on either side of suspected lesions. This technique has been validated in studies sponsored by HeartFlow, based in Redwood City, California, and the company now offers this service to cardiologists.
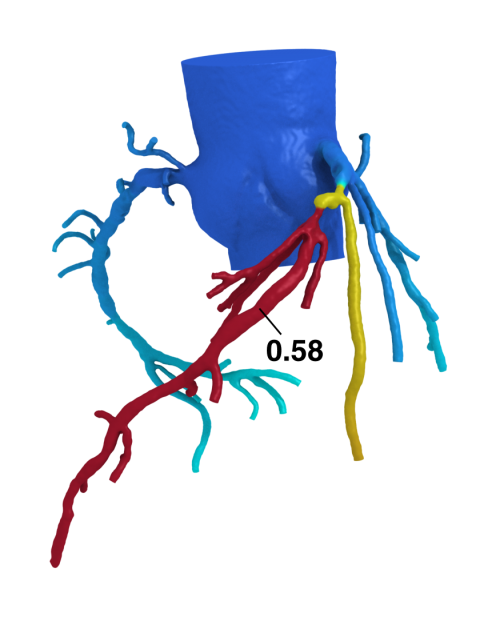
Going one step further, physicians at 11 heart centres in Europe and Asia initiated a study, led by Koo, to test a hypothesis that going backwards, they could look at CTA exams and identify not only which patients were at risk for a plaque rupture, but specifically identify the plaque that was going to rupture.
For the EMERALD study, sponsored by HeartFlow, Koo added computational fluids dynamics and fluid-structural interaction simulation to the FFR-CT calculations for 226 coronary plaques among the 71 patients enrolled. Emerald investigators first identified patient cases where the culprit plaque rupture had been documented using angiography, or intravascular imaging such as OCT or IVUS. Then the investigators searched the patient file to find a CT exam that had been performed ahead of the medical emergency. The average among enrolled patients was an exam performed one year earlier.
Emerald is an exploratory study and it shows we are very good at identifying the plaque at high risk for rupture eight times in 10, which is not just good but extraordinary.
John Stevens, HeartFlow CEO
These CT images were then run through the Emerald algorithms to assess plaque composition, lesion geometry and the haemodynamic forces. Results from the retrospective Emerald study were presented in May this year in Paris, at the interventional cardiology conference, EuroPCR. ‘Non-invasive haemodynamic data from CT was a better discriminator of lesions causal of ACS than stenosis severity or adverse plaque characteristics,’ Koo concluded.
Moving to a prognostic potential, Koo suggested the combination of all plaque characteristics might further improve the prediction of plaque-related clinical events to optimise treatment strategies for high-risk patients.
'Emerald is an exploratory study', said HeartFlow CEO John Stevens, ‘and it shows we are very good at identifying the plaque at high risk for rupture eight times in 10, which is not just good but extraordinary. For the moment we still have several hundred more patients cases for validation’.
Dr Pijls also presented his novel approach for measuring microcirculatory in myocardial irrigation at EuroPCR 2016, and when he had finished the panellists applauded in admiration and one of the audience step forward to say: ‘it blows my mind.’
The calculations for this novel cardiac quantification are so complex it took the software engineering of St. Jude Medical to capture them. The procedure, which required a controlled injection of saline solution into coronary arteries, is so complex that Paris-based Hexacath stepped in to invent a new catheter so that Pijls could realise his experiments.
Pijls then streamlined the procedure to conform to a standard cath lab workflow and removed dependence on operator interpretation. As a result, using the Hexacath RayFlow monorail infusion catheter combined with one of St. Jude’s pressure wires for FFR, and then standing back to watch the results on the St. Jude monitor, an interventional cardiologist can measure volumetric blood flow directly in selective coronary arteries during cardiac catheterisation and simultaneously, with the same guide wire, calculate the absolute myocardial blood flow, collateral flow, and myocardial resistance. ‘We don’t know yet what it all means, but at least we have a method to measure it,’ Nico Pijls concluded.
25.08.2016