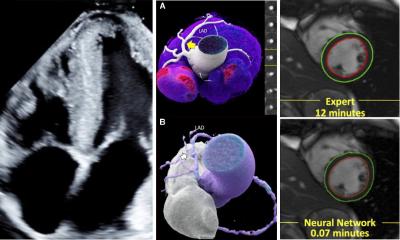
Every picture tells a story
Cardiologists are looking beyond on-screen images of the heart to extract data behind these scans that describe coronary dysfunctions and can reveal hard evidence of the cause of disease reports John Brosky.
Histology)
The advances in cardiac imaging are simply stunning.
We may be impressed with ever-more vivid displays of the breast, the spine, and even the brain, but no other organ holds the power to fascinate quite like watching the beating of a human heart.
Each year has brought breakthroughs for cardiac imaging from every branch of radiology technology, offering fresh and unexpected views.
Increased processing power has accelerated real-time reconstructions of scanned images in computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.
In the space of a few years we have moved from static black-and-white film to video-like color projections, from the flatness of two-dimensions to full-volume, three-dimensional renderings that can be viewed from any angle.
Cardiologists, however, are a tougher audience to please.
Once assured the breath-taking image accurately describe the anatomy they know so well, these no-nonsense clinicians quickly want to slice up the displayed picture to extract the digital data behind it to uncover hard evidence of a disease condition they can plainly see.
At the recent meeting of the French Society of Cardiology in Paris, the title of one scientific session bluntly expressed the prevailing attitude of cardiologists: “Real time 3D echography. Real breakthrough or just pretty pictures?”
The French society noted the revolution in cardiac imaging was an underlying theme for their conference and the program dutifully provided updates in what are acknowledged as breakthroughs in CT, MRI and the emerging potential for molecular imaging.
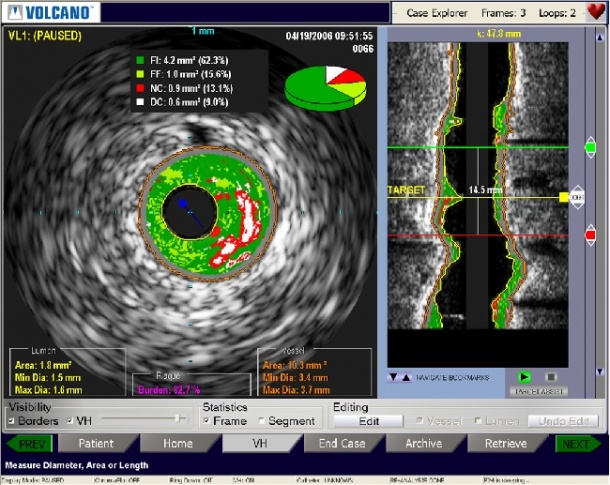
But after giving a nod to these high-powered modalities that are often beyond the reach of practicing clinicians, the weight of the program dedicated to imaging was heavily given over to sessions focused on two forms of cardiac ultrasound, echocardiography and intravenous ultrasound (IVUS).
Echo offers sound clinical data
Echocardiography is a familiar, readily available and relatively inexpensive means for non-invasive diagnosis, and with the introduction of had-held units in recent years, it has all but replaced the stethoscope for cardiologists.
Professeur Guillaume Jondeau from the Hôpital Bichat in Paris reminded colleagues that in 2008 practice guidelines of the European Society of Cardiology recommended echography as a first-line test in patients with a suspicion of cardiovascular disease, and that for a confirmation of a diagnosis, it is mandatory.
Beyond the immediate morphological and functional assessments essential to a rapid diagnosis and prognostic predictions, the critical, clinical advantage of echocardiography is a processing capability to rapidly translate the numeric bits and bytes into calculations of heart function.
Reviewing research in speckle-tracking echocardiography, Patricia Réant, MD, from the University Hospital Center in Bordeaux described how the distance between acoustic markers traced during a cardiac cycle can be used to calculate strain rates, or deformation, on three axis and that the global longitudinal strain (GLS) is providing new insights into coronary pathology.
GLS is proving to be a superior measure, she said, for understanding therapeutic effects of drugs and chemotherapies, and for assessing patients as candidates for cardiac resynchronization therapy (CRT).
“We are anticipating great enhancements to our understanding with the arrival of the new technologies for 3D strain analysis,” she said.
Eric Donal, MD, with the Pontchaillou University Hospital Centrein Rennes, is a co-author with Dr. Réant for a multicenter study that employed multiple parameters from echocardiography to increase the responsiveness of patients to CRT.
While agreeing 3D echo will create greater possibilities, he added that advances in cardiac MRI, in full volume cardiac CT scans, and in molecular imaging not only create new kinds of images, but add new forms of information as well.
“We are seeing very nice pictures,” he said, “and certainly these images greatly help us to communicate with physicians and colleagues.”
“Yet how useful these new technologies are for improving prognostics we can not say,” he said, “the new information needs to be considered a complement, and there is much work to be done.”
Light and sound in the same vein
Against the fresh views delivered from other technologies, the blurred black-and-white images of angiography look as out-of-date as photographs of grandmother in the family album.
Angiography may be losing its luster, yet it remains the undisputed standard for interventional cardiac navigation.
“It is the ambiguity of angiography that demands we go inside for a better view,” according to Gerard Finet, MD, an interventional cardiologist with the University Hospital in Lyon, who specializes in biomechanical factors for evaluating the risk of plaque rupture for the prestigious French national institute for medical research (INSERM).
Dr. Finet cites studies indicating that views of the interior structure of arteries using IVUS, or optical coherence tomography (OCT), positively influence major adverse cardiac events (MACE), such as death, myocardial infarction, stroke, or repeat revascularization.
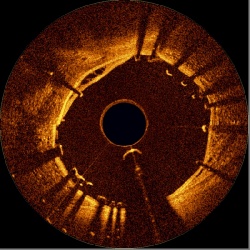
OCT is proving still proving its value for examining arterial structures, he said, but as it is dependent on light rather than sound, it lacks the penetration that IVUS affords for an in-depth examination of underlying tissue viability.
Pascal Motreff, MD, from the Univerity Hospital in Clermont-Ferrand agrees but demonstrated the superior visualization of arteries using OCT with a resolution 10 times greater than IVUS, and asserted this emerging modality is an essential complement to IVUS.
Using a series of traditional angiographic images, Dr. Motreff challenged the diagnoses offered by members of the audience by showing detailed examinations of the same lesions using OCT.
The nearly microscopic examinations possible with the new, high-speed generation of OCT enables a link-by-link review of stent placements providing for the first time an in vivo visualization of stent performance, such as stent under-expansion, which has been identified as a predictor of subacute thrombosis and restenosis.
04.03.2010