UTI pathogen
Boosting the Body’s Natural Ability to Fight Urinary Tract Infections
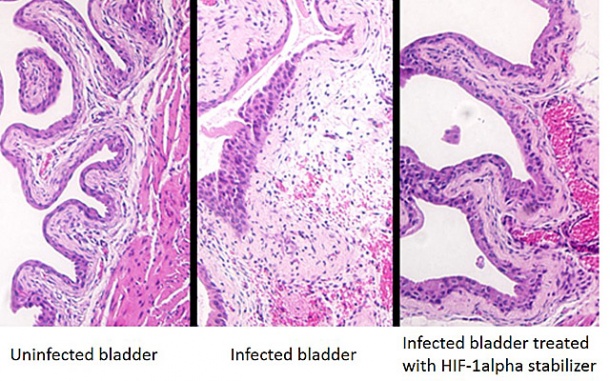
Urinary tract infections (UTIs) are common, and widespread antibiotic resistance has led to urgent calls for new ways to combat them. Researchers at University of California, San Diego School of Medicine and Skaggs School of Pharmacy and Pharmaceutical Sciences report that an experimental drug that stabilizes a protein called HIF-1alpha protects human bladder cells and mice against a major UTI pathogen. The drug might eventually provide a therapeutic alternative or complement to standard antibiotic treatment.
HIF-1alpha is known to influence the innate immune response, the body’s first line of defense against intruding pathogens. Like many regulator proteins, HIF-1alpha is relatively short-lived. To increase HIF-1alpha levels, researchers have developed drugs that delay its breakdown. This same pathway has been the target for drugs now in advanced clinical trials for treatment of anemia.
In this study, Victor Nizet, MD, professor of pediatrics and pharmacy, and colleagues explored the use of HIF-1alpha-stabilizing drugs to boost the innate immune response to uropathogenic E.coli (UPEC) bacteria, a major cause of UTIs. In healthy human urinary tract cells, treatment with the drugs increased HIF-1alpha levels. Such cells were then more resistant to UPEC attachment, invasion and killing than human urinary tract cells with normal HIF-1alpha levels.
Using a mouse model of UTI, the researchers showed that administration of HIF-1alpha stabilizers directly into the bladder protected against UPEC infection. They also found that invasion of bladder cells, a critical early step in the infection process, was reduced in treated mice compared to untreated mice.
To verify the importance of HIF-1alpha in the defense against UPEC infection, the researchers studied mice with reduced HIF-1alpha levels. Exposed to UPEC, these mice were more susceptible to bladder infection, and pre-treatment with HIF-1alpha stabilizers made no difference. This demonstrates that the drugs combat UTIs through their effect on HIF-1alpha.
Finally, the researchers examined whether treatment with HIF-1alpha stabilizers would be beneficial against an established UTI. To do this, they infected mice with UPEC first and then administered the drugs into the bladder six hours later. The treated mice had a more than 10-fold reduction in bladder colonization with the bacteria, demonstrating that HIF-1alpha stabilization is beneficial even after the initial infection.
“The ultimate goal of this research will be to advance HIF-1alpha stabilizers toward clinical trials in humans, using versions of the drug that can be taken orally and reach the urinary tract,” Nizet said.
Reference:
Co-authors include Ann E. Lin, Federico C. Beasley, Joshua Olson, and Nadia Keller, UC San Diego; Robert A. Shalwitz, Aerpio Therapeutics; Thomas J. Hannan, and Scott J. Hultgren, Washington University.
Source: UC San Diego
23.06.2015