4th International Multislice CT Symposium
Germany - The cost and user friendly processing of continuously increasing volumes of data, along with new of important technological innovations, were key issues during the 4th International Multislice CT Symposium, held this January in Bavaria.
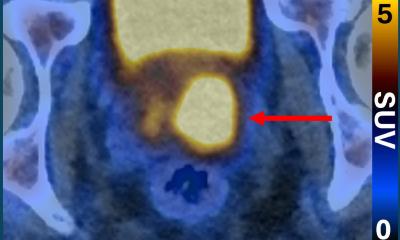
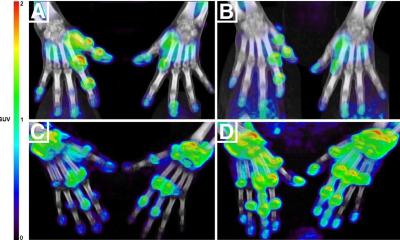
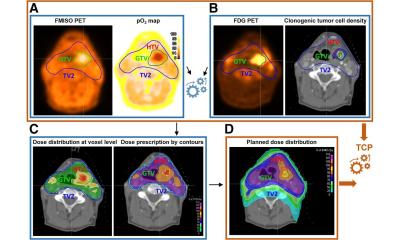
Combining multislice CT equipment with procedures used in nuclear medicine such as PET-CT (positron emission tomography) and SPECT-CT (single photon emission computed tomography) offers fascinating perspectives of capturing morphological image and functional diagnostics.

Due to the strong interest, we had to change the venue from Starnberg to the larger congress centre in Garmisch
Multi-modal examination procedures - particularly those used in molecular imaging - were among the key topics of discussions led by Professor Maximilian Reiser, Director of the Institute for Clinical Radiology at Munich’s Ludwig-Maximilian-University, and his colleague Professor Gary Glazer, President of the Department of Radiology at Stanford University, California.
Gary Glazer has witnessed the highs and lows of CT over 25 years. American physicist A M Cormack and British engineer G N Hounsfield built the first computed tomograph - independently of one another. In 1979 they shared the Nobel Prize for Medicine for this work. However, less than a decade later, CT looked like ‘old hat’. Then, in the early 1990s, Professor Willi Kalender, Head of the Institute of Medical Physics at the Friedrich-Alexander University, Erlangen, developed Spiral CT, and since the tube began to move continuously around the patient, scientists have improved CT enormously. Modern systems record picture signals with 64 detection channels, X-ray beam and detector rotate around the joint axis three times per second and achieve an isotropic, spatial resolution of 0.4 millimetres.
Further discussion revolved around the most recent development project at Siemens - the dual-source-CT, a system with two integrated tubes and detector systems. But the main topic was multi-modal imaging. By combining CT with a SPECT camera, or with positron emission tomography (PET), morphological and functional tissue information can be recorded simultaneously in one examination, and by superimposing both images radiologists can tell whether they are seeing a tumour or simply an infection.
‘Scientists aim to improve the spatial resolution of the systems to an extent where molecular structures become visible,’ Professor Glazer explained. ‘With CT we can only see tissue structures of several billion cells in size. However, for molecular imaging we need to make the smallest numbers of cells visible.’ An important question that molecular imaging hopefully will help to answer is: ‘What happens with stem cells injected into the heart? Do they remain in the heart and reproduce to the extent that they could possibly close holes in the cardiac wall?’ Ideally, we would be able to observe individual cell structures. Before cells start mutating into cancer cells they already have the relevant predisposition and send out abnormal signals. ‘Even the smallest tumours will have grown for several years.
To achieve successful therapy, diagnosis must be made at a much earlier time. Molecular imaging can show us how genes function and can even show single proteins or protein-protein interactions,’ Prof. Reiser pointed out. Researchers have been able to switch individual genes ‘on and off’ for some time in animal tests.
Individualised therapy is another aspect of molecular imaging. By precisely determining an individual patient’s genetic makeup, doctors hope to discover which patient responds to which medicine.
The development of cancer is hormone-dependent. For example, oestrogen levels significantly influence breast cancer. However, the metastases react in a different way to the primary tumour, so require different therapy.
Today, scientists can show individual genes and proteins. However, to make exact predictions they need to show several genes or proteins at the same time. This cannot be achieved with CT alone. The necessary resolution will only be achieved in combination with a modality that can also show cell functions.
Over the next 5-10 years there will be blood tests for many diseases or even for dispositions for diseases. Molecular imaging should help to achieve the change from ‘see and treat’ to ‘predict and prevent’. Professors Reiser and Glazer gave the symposium participants very interesting and fascinating insights into these topics so that many of them are already looking forward to the next symposium which is to be held in January 2008.
Details: www.radiologieforum.de
01.03.2006