Image source: Hokkaido University
News • Immune response regulation
Using iPS cells to fight transplant rejection
Scientists suggest a new strategy that uses induced pluripotent stem cells (iPSCs) to regulate immune reaction to transplanted tissues.
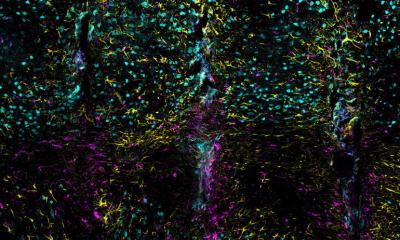
The team, led by Professor Ken-ichiro Seino of Hokkaido University’s Institute for Genetic Medicine, found that thymic epithelium cells derived from mouse induced pluripotent stem cells (iPSCs) can regulate immune response to skin grafts, extending their survival. The thymus, located behind the sternum, is a crucial organ for generating T-cells. T-cells control immune response, including organ rejection, and are closely associated with immunological self-tolerance, the ability of the immune system to recognize self-produced antigens as non-threatening.
The researchers published their findings in the journal Scientific Reports.
This finding will contribute to the advancement of regenerative medicine using cells and tissues derived from iPSCs
Ken-ichiro Seino
Pluripotent stem cells such as embryonic stem cells (ESCs) and iPSCs, which are capable of differentiating into various types of cells, are expected to be an alternative source of grafts for transplantation. But when an organ or tissue from a donor is transplanted, the grafts are rejected and eventually destroyed by the recipient’s immune system. The same holds true for cells or tissues derived from pluripotent stem cells. In regenerative medicine, it is therefore important to regulate immune reaction for transplants to succeed. While past research found that it is difficult to efficiently make thymic epithelium cells from iPSCs, the group discovered that the introduction of Foxn1, a pivotal gene in the thymus, to mouse iPSCs helps the efficient differentiation of such cells.
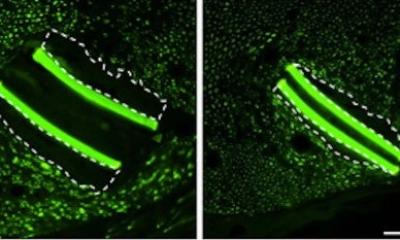
The team then transplanted to the recipient mice the thymic epithelium cells derived from iPSCs and skin grafts of genetically compatible donor mice. The results showed that the skin grafts in the recipient mice survived longer when iPSC-derived thymic epithelium cells were transplanted in advance. “This suggests it is possible to regulate immune rejection by transplanting immune-regulating cells derived from iPSCs before conducting cell or tissue transplants. This finding will contribute to the advancement of regenerative medicine using cells and tissues derived from iPSCs,” says Ken-ichiro Seino of the research team.
Source: Hokkaido University
07.02.2020