Article • Real-time radiation plans, X-core MRI
Significant advances in medical physics
To target ionised radiation as precisely as possible, imaging a tumour is vital in radiotherapy planning. ‘Today, imaging is used increasingly during the therapy itself,’ explained Professor Mark Ladd, Head of Medical Physics in the Department of Radiology at the German Cancer Research Centre (DKFZ) in Heidelberg, Germany, and President of the German Society for Medical Physics (DGMP), during the society’s 51st annual meeting.
Interview: Daniela Zimmermann

This new imaging role is possible due to the magnetic resonance linear accelerator, MR-Linac for short, which combines a linear accelerator with a MRI scanner. ‘It shows, in real-time, how the tumour moves with breathing,’ Ladd explained during our interview regarding current trends in medical physics.
Continuous MR-Linac scanning during radiotherapy enables tracking of breathing-induced movement and thus adjust the therapy; it also records any change in location and size of the tumour during the weeks of therapy. ‘Rather sooner than later, we’d like to control the collimators in real-time so as to adjust radiation in view of the changes, compared to the initial therapy plan. In short: we want to tailor the therapy plan in real-time,’ Ladd underlined. The more precisely the tumour can be targeted, the higher the dose can be. ‘Thus the patients wouldn’t have to undergo 30 radiation sessions but maybe only five,’ he pointed out. At DKFZ a project-team is looking for new approaches to adjust the collimators quickly.
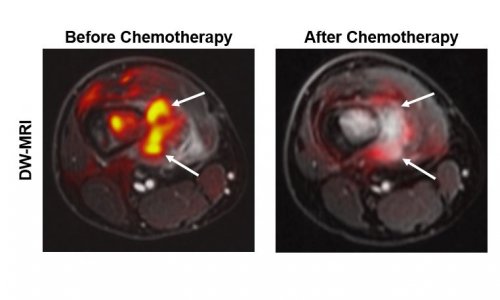
Image source: Daniela Thorwarth / University Hospital Tübingen
Another research project in particle therapy deals with the exact recognition of the Bragg peak, i.e. the point when the energy of the ion beam reaches its peak and then sharply decreases. Since scientists are still uncertain where exactly the Bragg peak ends in the body, a comparatively large safety margin is defined around the tumour to reach as much of the tumour tissues as possible. ‘With current technology the safety margin has to be so large that, in fact, we cannot realise the full potential of proton therapy,’ Ladd reported. Thus, researchers are trying to develop different methods to determine the Bragg peak in vivo. One of these approaches tries to detect secondary gamma rays triggered by the proton beam in the body.
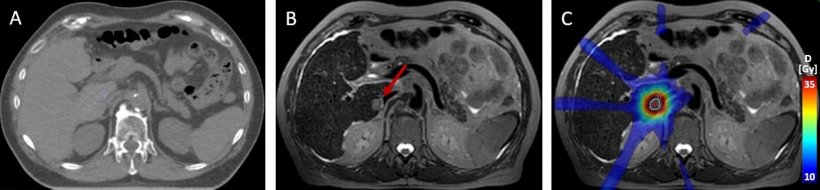
Image source: Daniela Thorwarth / University Hospital Tübingen
Better insights: photon-counting CT
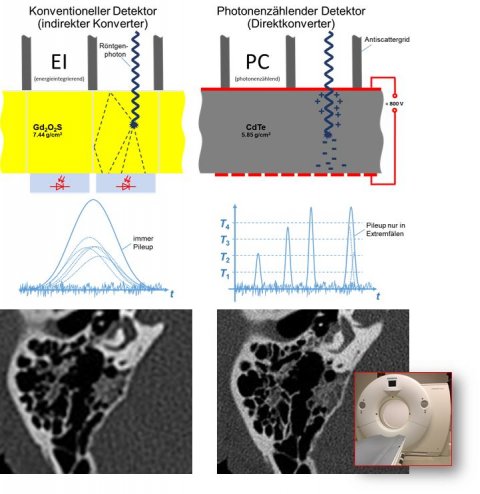
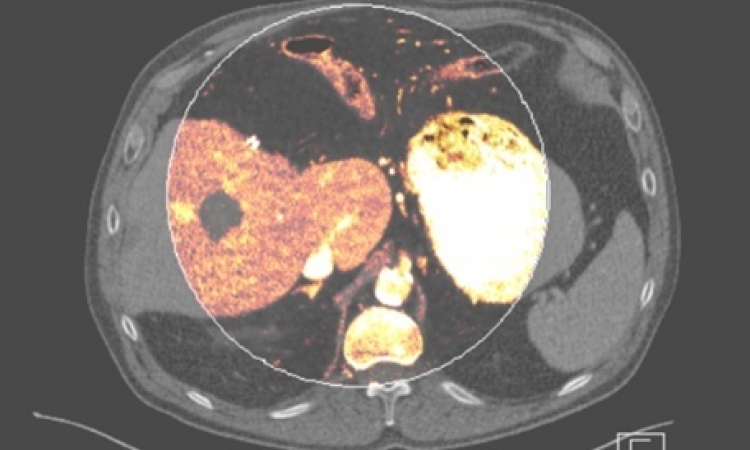
Image source: DKFZ/University Hospital Heidelberg
Photon-counting CT is another remarkable development in radiology. DKFZ houses one of three CT prototypes worldwide that feature a unique component: a photon-counting detector made of a semi-conductor material that can directly convert X-rays into electrical signal impulses, making it particularly efficient. ‘I do hope that this technology will one day be clinical routine,’ Ladd said.
While photon-counting detectors are expensive, they offer better resolution and allow imaging with significantly lower contrast doses. ‘With regard to the current debate about contrast media, photon counting is a very promising approach,’ he said.
MRI with new elements and high field strength
Another trend in medical physics is X-core MRI. While in conventional MRI the spin of the hydrogen nucleus is measured, X-core MRI determines the magnetic moment of isotopes of other nuclei, e.g. sodium-23 (Na-23), oxygen-17 (O-17), potassium-39 (K-39) or chlorine-35 (Cl-35). Problem: these isotopes are rather rare. Only 0.04 percent of oxygen, for example, is O-17. In a DKFZ research project, patients inhale a gas enriched with O-17 during the MRI scan. The oxygen travels through the blood and as soon as it reaches the mitochondria in the cells and is converted into water it can be visualised. ‘This enables us to precisely measure the local oxygen metabolism in the brain and in other parts of the body,’ Ladd explained. Currently, one team is trying to detect whether regional differences in metabolism might indicate which areas of the brain will recover after a stroke and which ones won’t.
Recommended article

Article • X-Nuclei MRI
Oxygen provides insights into tumour metabolism
Magnetic resonance imaging (MRI) usually measures the magnetic moment of the hydrogen atomic nuclei arising from the spin. However, scientists at the German Cancer Research Centre (DKFZ) are investigating the spin of other nuclei for imaging: ‘X-nuclei imaging has a large potential for MRI imaging as the x-nuclei play an important part in many physiological processes,’ according to doctor and…
Visualising the distribution of O-17 and other isotopes in MRI requires a field strength of 7-Tesla. Indeed, beyond this application 7-T MRI is one of the key development areas in medical physics. Since 2017, the first systems have been certified for clinical exams, initially limited to head scans and smaller joints. The advantage of a 7-T MRI scanner is not only resolution. ‘There is an interdependence of sensitivity and time.’ Ladd said, adding ‘with a field strength of 1.5-T we can examine about everything that we can examine with 7-T – but at a much slower speed.’
3-T scanners are mostly used to perform exams faster than with a 1.5-T scanner. By contrast, a 7-T scanner not only reduces exam time but also offers enhanced spatial resolution and more detail. “You can compare it with standard and HD resolution of a TV screen: suddenly you see things you could not see before,’ Ladd said. For example, the so-called swallow tails in the basal ganglia, whose lack indicates Parkinson’s disease, can only be vaguely seen in a 3-T scan – but they are clearly visible in a 7-T scan.
Gradient coils: pushing the envelope of physics
Whole-body PET-CT could open up new possibilities in imaging, which were unrealistic before due to the exceedingly high radiation exposure
Mark Ladd
Ladd sees larger gradient fields as an overall trend in MRI. The faster the gradient, the higher the risk of peripheral nerve stimulation. This fact has limited the use of larger gradient fields. GE Healthcare, however, has developed a 3-T system for the head only, which is equipped with a faster gradient since, in head-only exams, the problem of nerve stimulation is less pronounced.
Some research teams have managed to simulate nerve stimulation caused by gradient coils prior to the actual scan. Before, the degree of nerve stimulation was measured using fully developed gradient coils and a performance cap for this particular coil was determined. Today, computer programs can predict the degree of peripheral nerve stimulation, thus the coils can be optimised in the development phase. ‘Over the next few years, we will see significantly stronger gradient systems,’ Ladd predicts.
PET-CT and the 2 metre detector
Last, but not the least in PET-CT, the first whole-body scanner may be in the wings. The Chinese company United Imaging developed a detector that measures two metres! Siemens is working on a system that combines four conventional detectors and thus enables a PET-CT scan of more than a metre. ‘Whole-body PET-CT could open up new possibilities in imaging, which were unrealistic before due to the exceedingly high radiation exposure,’ Ladd explained, ‘including new options for visualising the course of a therapy.’
Profile:
Having joined the German Cancer Research Centre (DKFZ) in Heidelberg in 2013, today Professor Mark Ladd heads Medical Physics in its Department of Radiology. He is also President of the German Society for Medical Physics (DGMP). His research focus is magnetic resonance imaging (MRI), particularly the launch and further development of new methods using ultra-high-field strengths and MRI in image-guided radiotherapy. He studied electrical engineering at the University of Michigan in Ann Arbor and Stanford University in California. In 1998, and received his doctorate from ETH Zurich in the context of a research cooperation project between the University Hospital Zurich and GE. In 2004 he became Professor of Biomedical Imaging at Essen University Hospital, where he increasingly focused on ultra-high-field MRI.
03.12.2020