Image source: Adobe Stock/happyvector071
News • Novel approach in pathology
Laser microbiopsy: a minimally invasive method to extract tissue samples
Biopsies are one of the most widely used diagnostic procedures for many types of diseases, including cancer.
Today, the gold standard is to mechanically extract a tissue section, cut it into thin slices, and stain these with hematoxylin and eosin (H&E) to make the nuclei and cytoplasm of cells easier to see with a microscope. While this approach is well established in clinical practice, it has several disadvantages that call for the development of alternative procedures.
In a recent study published in Journal of Biomedical Optics, a research team led by James W. Tunnell of The University of Texas at Austin, and Thomas E. Milner of University of California Irvine, developed a revolutionary laser-based approach to perform microbiopsies. Their method involves using a concentrated laser beam to eject a small volume of cells from the irradiated tissue, collect them onto a glass slide, and use a staining procedure called virtual H&E. “The traditional pathology workflow suffers from limitations, including biopsy invasiveness and complex and time-consuming tissue processing; our protocol based on laser microbiopsy with virtual H&E imaging shows promise as a rapid and minimally invasive tool for biopsy and diagnosis,” highlights Jason B. King of The University of Texas at Austin, first author of the study.
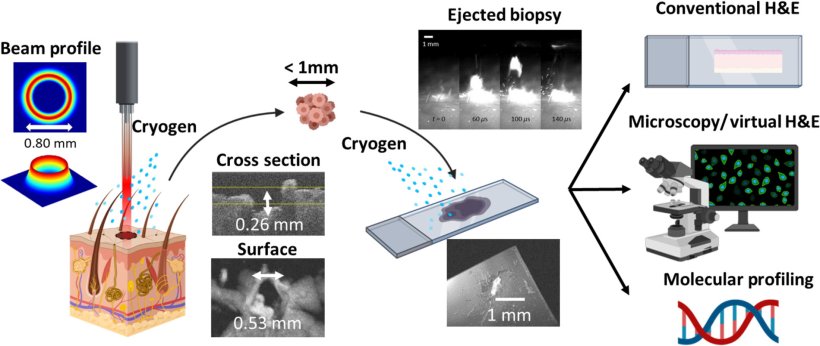
Image source: King et al., Journal of Biomedical Optics 2022 (CC BY 4.0)
The main mechanism at play in this novel microbiopsy technique is laser ablation. This is a phenomenon in which the focused energy of laser pulses is absorbed by the target throughout a small superficial volume, causing matter to quickly heat up and be ejected. Although laser ablation is more widely used in machining and materials manufacturing, the researchers sought to use this principle in a manner compatible with tissue extraction and analysis.
To achieve this, the team had to overcome several obstacles. First, they had to design the optical setup and optimize the energy and profile of the laser beam. Using only commercially available equipment and an FDA-approved Ho:YAG laser, the researchers built a benchtop laser microbiopsy system that would enable convenient use in a clinical setting. After carefully analyzing the characteristics of the beam profile theoretically and through experiments, they performed full-fledged computational simulations of the entire laser microbiopsy process. This provided valuable insight into how the laser beam could interact with tissue and allowed them to select the best parameters before proceeding to actual experiments.
Once a prototype was ready and configured, the researchers used the system to extract extremely small volumes of tissue (below the microliter range) from porcine skin and kidney. They stained these tissue samples for virtual H&E and analyzed them with a confocal microscope. One important aspect of the proposed protocol is that the researchers sprayed both the target tissue area and the glass slide with a cryogenic spray to limit the extent of thermal damage caused by the ablation process. This proved to be essential to obtain good quality samples.
The overall results of the experiments are very promising, as most of the sampled tissue was free from thermal damage. The extracted volumes were low enough to be minimally invasive, yet sufficient for virtual H&E analysis, as well as other analytical techniques such as traditional H&E and protein or genetic testing methods. This means that a patient could undergo dozens of laser microbiopsies without causing nearly as much harm as standard biopsies.
Another remarkable advantage is that the entire tissue extraction and virtual H&E analysis protocol can be completed in only few minutes, as opposed to half an hour or even days for standard biopsies with conventional tissue processing. To top this off, there is no need for a highly trained technician nor expensive microscopy equipment beyond a confocal microscope.
These qualities make the proposed laser microbiopsy system an attractive first-pass screening tool to determine if a full-scale biopsy is needed. The research team also believes that this method could be used during surgery to sample tissue from delicate structures and organs while preserving as much healthy tissue as possible. This may be possible through further refinement of the system’s optics and size.
Source: SPIE
16.12.2022