Video • Coronavirus medication research
Lab-grown beating heart cells could identify Covid drugs
Scientists have grown beating heart cells to attempt to identify drugs to prevent Covid-19-related heart damage.
Report: Mark Nicholls
Concerns over the extent of cardiac damage among Covid patients emerged during the coronavirus pandemic and there are also suggestions that the impact on cardiomyocytes could contribute to the symptoms of long Covid. To explore these issues, a research team at the University of Cambridge used the lab-grown beating heart cells to demonstrate how effective the experimental peptide drug DX600 was in preventing SARS-CoV-2 entering the heart cells.
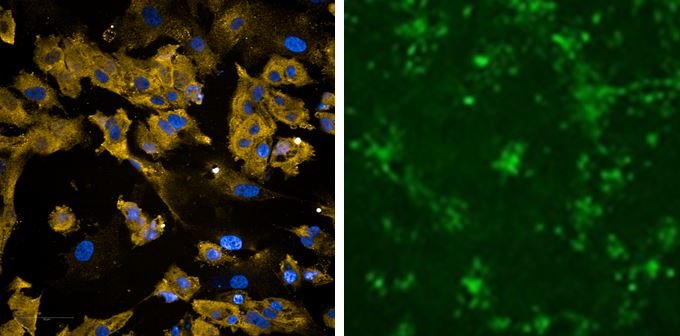
Right image: heart cells infected with SARS-CoV-2 shown in green
Image source: Williams et al., Communications Biology 2021 (CC BY 4.0)
For the study, which was published in the Nature journal Communications Biology, the Cambridge scientists used human embryonic stem cells to grow clusters of heart cells, which also contained the ACE2 receptor. SARS-CoV-2 hijacks the ACE2 protein on the cells to allow the virus to enter cells and spread. Researchers used a modified synthetic virus decorated with the SARS-CoV-2 spike protein to mimic how it infects heart cells and then used the model to screen for potential drugs to block infection. They showed that some drugs that targeted the proteins involved in SARS-CoV-2 viral entry could significantly reduce levels of infection, including an ACE2 antibody that has been shown previously to neutralise pseudotyped SARS-CoV-2 virus, and the experimental drug DX600, which is an ACE2 peptide antagonist.

While it is well-documented that the heart is one of the main organs damaged by the virus, scientists remain unsure of the exact mechanism causing the damage. Professor Anthony Davenport from the Department of Medicine and a fellow at the University of Cambridge, who led the study, said the results blocking the viral spike binding to ACE2 supports the concept that ACE2 is the viral entry receptor in human cardiomyocytes. Having shown that infection by SARS-CoV-2 was dependent on the concentration of the virus surrounding the cardiomyocytes, researchers believe that blocking entry of the virus to reduce the viral load that the body is exposed to should reduce cell damage.
He said: “We need to do further research on DX600 it could provide us with a new treatment to help reduce harm to the heart in patients recently infected with the virus, particularly those who already have underlying heart conditions or who have not been vaccinated. We believe it may also help reduce the symptoms of long Covid.” This is of significance as there is evidence to suggest that patients with underlying heart condition are more than four times as likely to die from Covid-19.
We have the potential to apply precision medicine to control the virus by combining this test with novel drugs
Anthony Davenport
This could also help advance research into the impact and treatment of coronavirus, he said. “We currently have one of the most accurate, rapid and widely available companion diagnostics – the PCR test - to identify individuals infected with SARS-CoV-2. We have the potential to apply precision medicine to control the virus by combining this test with novel drugs.” But he added that new medicines are required for prophylactic treatment for those where vaccination against SARS-CoV-2 is contraindicated, and to address the unmet need to protect the heart against acute damage during hospitalization, as well as during the prolonged post-viral recovery.
With DX600 now shown to inhibit binding of the spike protein to ACE2, he said that studies to show it is effective in blocking viral entry in animal models are need in order to begin translation into the clinic. Professor Davenport said: “Our results provide a platform that has passed proof-of-principle to generate high quality data in pre-clinical studies to justify translational research in animal models of acute respiratory distress syndrome and the repurposing of current drugs for clinical trials.”
The research with the beating heart cells – supported by Wellcome, Addenbrooke’s Charitable Trust, Rosetrees Trust Charity and British Heart Foundation - could potentially open the door to other applications. “We have successfully grown clinically relevant beating heart cells in 96-well plates which will enable pharmaceutical companies to carry out high throughput screens of thousands of compounds using automated platforms,” said Professor Davenport. “Screens for new medicines can be refined by genetically engineering cardiomyocytes to mimic defects in specific proteins that cause diseases such as Marfan syndrome.”
Profile:
Anthony Davenport is Professor of Cardiovascular Pharmacology at the University of Cambridge and Director of the Human Receptor Research group. A key aim of his research interest lies in helping to understand the role of G-protein-coupled receptors (GPCRs), together with their transmitters, in the human cardiovascular system and how these are altered with disease, in particular the consequences of endothelial cell dysfunction, to identify new targets for novel drugs.
27.08.2021