Image source: Rolf Müller/Universitätsklinikum Bonn (UKB)
News • Chemotherapy, testicular cancer
Gene scissors find target for testicular cancer therapy
Bonn researchers uncover contribution of protein degradation processes to cisplatin resistance in germ cell tumors: Cisplatin is used successfully in the chemotherapy of testicular cancer. However, patients who develop resistance to the cytostatic drug urgently need alternative therapy options.
Researchers at the University Hospital Bonn (UKB) have now been able to elucidate a mechanism underlying cisplatin resistance in testicular cancer. Using CRISPR gene scissors, they identified the NAE1 gene as its driver. Inhibiting this resistance mediator by adding the NAE1 inhibitor MLN4924 not only restores the effect of cisplatin, but also has an additional killing effect on tumor cells. The study results have recently been published in the British Journal of Cancer.
Testicular cancer, also known as testicular germ cell tumors (TGCT), is the most common type of cancer in young men. When treated with cisplatin, the tumor cells stop growing and die. This is because the cytostatic drug causes damage to DNA, the carrier of genetic information, thus stopping the cell cycle. "The alarm signal is sent 'Attention, repair, do not divide further.' To do this, the cell needs, among others, so-called tumor suppressor genes that put the brakes on precisely this kind of damage and do not release cell growth until it has been repaired," says Prof. Hubert Schorle from the Institute of Pathology at UKB. The chances of cure with this type of chemotherapy are exceptionally high for testicular cancer. However, in some patients the tumor develops cisplatin resistance, which is associated with a reduced survival rate.
To get to the bottom of the causes of cisplatin resistance in testicular cancer, the Bonn research team used CRISPR gene scissors. They used them to activate each gene once in testicular cancer cells. They then treated the genetically modified cell culture with cisplatin and singled out those cells that had survived and in which, as a result, the cytostatic drug was no longer effective. "By analyzing the DNA of these cells, we found where the gene scissors were active and were able to identify the genes responsible for cisplatin resistance. In addition to genes already known for cisplatin resistance, to our surprise we found the gene NAE1, the regulator of the neddylation cascade," says first author Kai Funke, a PhD student of Prof. Schorle .
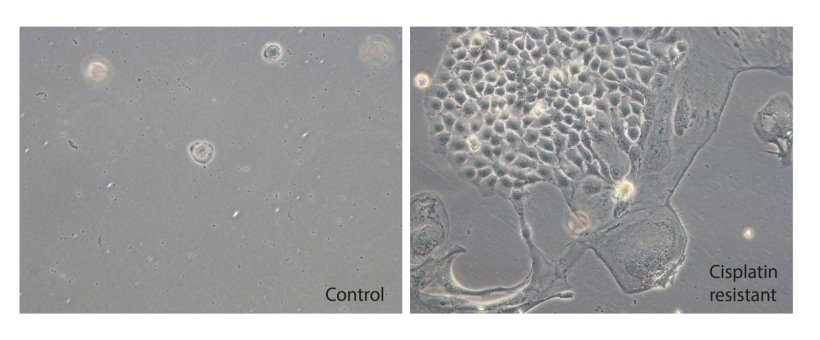
Image source: Kai Funke/Universitätsklinikum Bonn (UKB)
A cell must be able to precisely control the type and amount of different proteins. In addition to the regulation of protein production, control over the targeted degradation of proteins is also important. Here, the neddylation cascade marks the proteins destined for degradation. "It appears that tumor suppressor proteins, which act as brakes on cell growth in the presence of DNA damage, are an important target of neddylation. Accordingly, they are degraded to a greater extent and their braking effect is lifted when the neddylation cascade is overactive due to the upregulated gene NAE1," explains Prof. Schorle, who is also a member of the Transdisciplinary Research Area (TRA) "Life and Health" at the University of Bonn.
In turn, inhibition of the neddylation cascade leads to an accumulation of suppressor proteins and, consequently, to a halt in cell division. With addition of the NAE1 inhibitor MLN4924, the Bonn researchers therefore observed a re-sensitization to the cytostatic drug in cisplatin-resistant testicular cancer cells. The fact that connective tissue cells were not affected by the treatment gives hope that there might be only weak side effects when applied. Thus, the Bonn researchers highlight the additive effect of NAE1 inhibition by MLN4924 in combination with cisplatin as a new treatment option for testicular cancer. "We describe here for the first time neddylation as a therapeutic target in testicular germ cell tumors. Inhibition of neddylation is already being investigated in other tumor types in clinical trials. In pancreatic cancer MLN4924 has also been shown to have an additive effect with cisplatin," says Prof. Schorle.
Source: Universitätsklinikum Bonn (UKB)
13.04.2023