Image source: Fdardel, Aptamer biotin, CC BY-SA 3.0
Article • Antibiotic resistance
Five-minute MRSA detection with aptasensor swabs
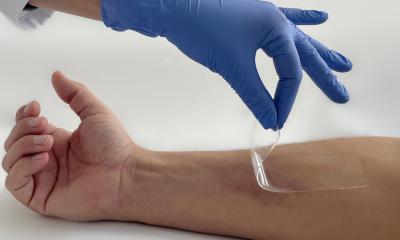
An international research team from Saudi Arabia, Germany, and Jordan has developed a novel pathogen aptasensor swab designed to qualitatively detect, within five minutes, any methicillin-resistant Staphylococcus aureus (MRSA) contamination that remains in a hospital isolation room or other surface following standard decontamination and cleaning.
Aptasensors are small, synthetic nucleic acid-based affinity binders with high selectivity and specificity to a specific target. They are easy and inexpensive to mass produce and have very little variation from batch-to-batch. ‘Our engineered aptasensor targets MRSA selectively and does not bind to any other bacterial pathogen on a multi-contaminated surface,’ explain the researchers in Biosensors and Bioelectronics (issue: 15/3/2021). ‘We believe the engineered aptasensor holds great promise for the rapid and multiplexed detection of MRSA and other life-threatening infectious pathogens in the hospital environment.... It offers a means to rapidly detect residual viable bacteria, such as MRSA, on frequent touch surfaces that may have survived after being cleaned.’
The researchers explain that aptamers, oligonucleotide or peptide molecules that bind to a specific target molecule, have important advantages compared with antibodies. In addition to being easier and cheaper to mass produce or modify, and without batch-to-batch variation because they are synthetically produced. They can be incorporated into assays that need heating because they have better thermal stability. Additionally, they can better detect small, poorly immunogenic biomolecules independent of carrier molecules.
MRSA contaminated swabs turned from white to blue

For the validation study, principal investigator Professor Mohammed Zourob PhD, and co-researchers, prepared sterile cotton swabs that were infused with MRSA-targeting aptamers. The swabs were placed in tubes containing MRSA of differing concentrations and a sterile control tube for three minutes and then washed with a phosphate-buffered saline (PBS) solution. Testing was also performed on glass slides and plastic containers, but no absorbable surfaces. Swabs contaminated with MRSA changed from white to blue, with more vivid colour correlating with greater levels of MRSA. Qualitative and quantitative analysis showed that MRSA was detected in the concentration range of 102 to 108 CFU/ml.
Because the cotton swab serves as both a collection and concentration tool, it eliminates the need to be sent to a lab for both enrichment and culture. The rapid turnaround time enables users to identify MRSA rapidly, before another patient or staff come in contact with the contaminated surface and to prevent spread and cross-contamination. It also is a quality control tool to show if more stringent environmental cleaning protocols are needed.
Zourob told Healthcare in Europe that real-world validation is being planned in several hospitals in Saudi Arabia. ‘We’ll conduct this study on actual patient samples in hospital environments. We expect this validation procedure to take approximately six months.’ (CK)
01.07.2021