European nanomedicine research
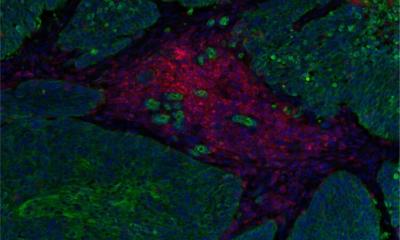
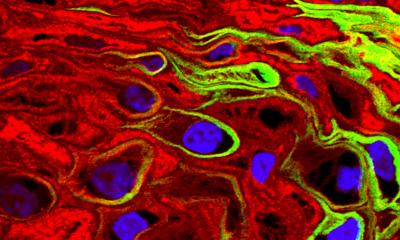
Cancer diagnostics - Nanotechnology is currently being used in oncology to improve early tumour detection, imaging procedures and targeting of cancer therapies. Cancer biomarkers, indicators that are being produced by the body in spreading tumour cells, play an important role in cancer detection, Dr. Jörg Raach reports
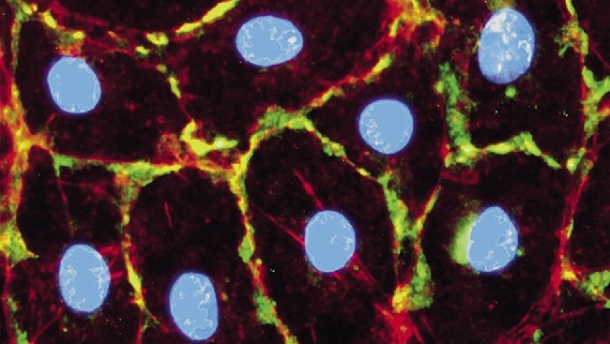
However, in the early stages of the disease, due to their concentration in the body, these markers are difficult to detect safely and efficiently. The targeted delivery of nanoparticles in the suspected tumour area can prompt the cancer cells to increase the production of biomarkers significantly. Consequently, the biomarkers are detected more easily and cancer can be diagnosed much earlier than with a biopsy. This in turn means that a suitable therapy can be initiated in this early stage of the disease that is less stressful for the patient and very likely provides better outcomes.
Iron oxide nanoparticles can be coated with a special substance that docks easily onto tumour cells. Due to their magnetic properties they are particularly well suited for MRI scans. Their se and concentration in the tumour, as visualised in the MR image, facilitate precise mapping of lesions and allow the surgeon to better plan tumour resection.
Therapy - In cancer therapy nanotechnology is employed for targeted drug delivery and basic therapies. Nanoparticles can be injected into a tumour and activated by magnetic fields, X-rays or light to produce heat in a defined area. Thus tumour cells can be destroyed without harming surrounding tissue. A second strategy to deliver substances in oncology is encapsulating chemotherapy agents or genes in nanoparticles. This reduces the amount of necessary medication significantly and also diminishes adverse effects. Both methods have been successfully coupled with gold nanorods already; they transport drugs to the tumour where they are stimulated by infrared light. The heat triggers the drug release and supports cancer cell destruction.
Fourteen companies are already marketing 77 nanomedicine products for cancer therapy and 82 clinical trials are underway.
The research network - Founded in 2005, The European Technology Platform Nanomedicine (ETPN) is an EC initiative along with large companies (inter alia Philips, Siemens, UCB), SMEs and research institutions to conduct medical nanotechnology research and promote joint projects. Since its inception ETPN has published several strategic documents that define requirements and roadmaps for research in nanomedicine. ETPN is also involved in many EU-funded projects that aim to create a suitable social and economic environment as well as the structural preconditions for the efficient translation of research and development into innovative nanomedicine products (see www.etp-nanomedicine.eu).
Since 2012, the NanoMed 2020 project has been interlinking around 200 European research institutions, hospitals and firms to further explore the potential of nanomedicine.
The 18-months project receives €500,000 in EU funding.
18.11.2013