Image source: Willer et al., The Lancet Digital Health 2021 (CC BY 4.0)
Article • Modified radiography technique
Dark-field X-ray casts new light on lungs
It looks like a negative of a normal X-ray image but gives new insights into pathologies of the lungs and other organs: dark-field radiography uses a part of X-rays that has hardly been considered so far to expand the spectrum of diagnostic imaging. At the ECR congress, Theresa Urban from the Technical University of Munich (TUM) presented the new method and explained its advantages in the diagnosis of emphysema.
Report: Wolfgang Behrends

Photo: supplied
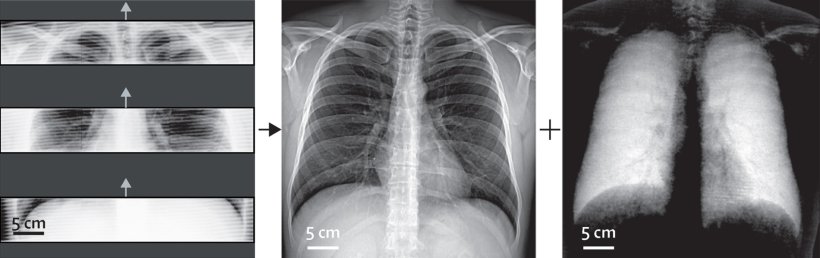
‘Unlike conventional radiography, which produces images by penetrating anatomical structures to varying degrees, dark-field imaging is based on the small-angle scattering of X-rays,’ the expert explained. ‘This effect occurs at transitions between materials with different densities, for example between tissue and adjacent air or fluids. The principle is similar to light refraction in water, only on a much smaller scale.’ The evaluation of this small-angle scattering provides information on microstructures that cannot be seen in normal X-rays.
To generate the dark-field images, an interferometer is coupled to the X-ray device to record the scattering and translate it into image information. ‘A major advantage is that the image information from conventional and dark-field X-rays is recorded in parallel,’ Urban explained. This way, both modalities are available in a complementary way for diagnostics without the need for multiple radiation exposure.
Insight into microstructures improves early detection
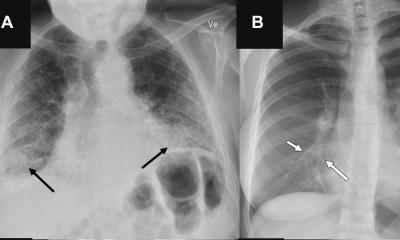
So far, the lung has been the primary field of application for the new imaging technique. The organ offers good conditions for dark-field radiography because its alveolar structure contains numerous transitions between air and tissue. ‘In a healthy lung, these structures are intact, thus producing a strong dark-field signal. On the images, these areas show up brightly. In contrast, emphysema creates dark patches; it destroys the alveolar structure so that large air bubbles appear with only few transitions.’
Normal X-ray imaging can also detect such pathologies in the lungs, but at a much later stage, Urban pointed out. ‘With dark-field radiography, there is no superimposition of bones and surrounding tissue, so that we get a clearer view of the microstructure and pathological changes can be detected earlier.’ Thus, pulmonary emphysema can already be seen in the early stages,1 while conventional X-ray imaging only registers anything upward of moderate manifestations.2,3 ‘Up to now, CT imaging has been the method of choice for early detection, but dark-field imaging offers much lower radiation exposure while maintaining comparable sensitivity.’
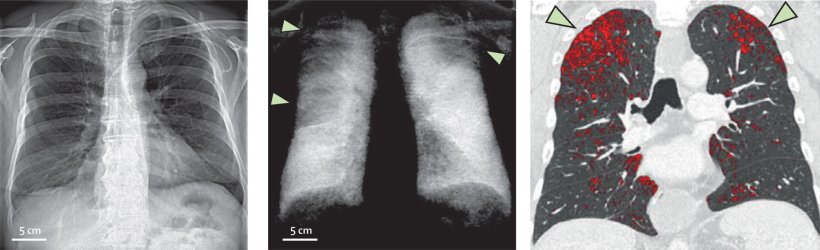
Image source: Willer et al., The Lancet Digital Health 2021 (CC BY 4.0)
Research is currently being conducted to find out whether the new method can also provide added diagnostic value for other lung diseases – with Covid-19 being an obvious focus; so far, the results of the studies are very promising, the expert said.
The use of dark-field X-rays is by no means limited to the lungs, explained Urban: ‘In principle, the modality is suitable for all areas with a microstructure that is changed or arises as a result of a disease.’ This applies, for example, to the formation of kidney stones, microcalcifications in mammography or musculoskeletal diseases such as osteoporosis, all of which can be seen in the dark-field. It is also conceivable to use it to visualise uric acid crystals that form in gout disease.
Three-dimensional advances
Several important milestones have already been mastered on the way to clinical application: In October 2018, the first clinical trial for the early detection of COPD started, with results recently published in the journal Radiology.4 ‘We have already proven the added value of dark-field imaging in emphysema and are confident that we will also succeed in other clinical applications.’
Ongoing dark-field research also includes the combination with other imaging modalities such as CT.5 Using cross-sectional slices, the information gathered from the dark-field can be translated into three-dimensional images. However, using CT poses additional technical challenges for the scientists. For example, the rotation of the scanner’s gantry causes vibrations that must be subsequently calculated out to obtain a viable dark-field image. First measurements on thorax phantoms were successful, so that dark-field CT will soon be used on patients.
A major benefit of dark-field imaging is that the method is compatible with commercially available X-ray systems, only an interferometer needs to be added. The company that manufactures the microgrids used in these devices is already working on bringing conversion kits to market, Urban reported. ‘We hope that dark-field technology will become widely accepted and that many patients will be able to benefit from it.’
Profile:
Theresa Urban, MSc, is a PhD student at the Department of Biomedical Physics at the Technical University of Munich (TUM) and the Munich Institute for Biomedical Engineering. She is part of Prof. Dr. Franz Pfeiffer's Biomedical Physics group, which focuses on the translation of modern X-ray concepts into clinical applications.
References:
- Defined after: Lynch et al., Radiology 2015: CT-Definable Subtypes of Chronic Obstructive Pulmonary Disease: A Statement of the Fleischner Society; https://doi.org/10.1148/radiol.2015141579
- Willer et al., The Lancet Digital Health 2021: X-ray dark-field chest imaging for detection and quantification of emphysema in patients with chronic obstructive pulmonary disease: a diagnostic accuracy study; https://doi.org/10.1016/S2589-7500(21)00146-1
- Urban et al., Radiology 2021: Qualitative and Quantitative Assessment of Emphysema Using Dark-Field Chest Radiography; https://doi.org/10.1148/radiol.212025
- Gassert et al., Radiology 2021: X-ray Dark-Field Chest Imaging: Qualitative and Quantitative Results in Healthy Humans; https://doi.org/10.1148/radiol.2021210963
- Viermetz et al., PNAS 2022: Dark-field computed tomography reaches the human scale; https://doi.org/10.1073/pnas.2118799119
13.07.2022