Coronary artery disease in diabetes mellitus
By Dr Akhil Kapur of the London Chest Hospital, United Kingdom
Recent advances in percutaneous coronary intervention (PCI) may offer a new approach to treating this complex group of patients.
Diabetes mellitus is strongly associated with the development of coronary artery disease (CAD) - up to one third of patients attending cardiac catheterization laboratories have diabetes. In addition, treatment outcomes are significantly poorer in this group of patients compared with the general population (restenosis rates [RR] are higher and clinical outcomes are worse). Thus patients with CAD and diabetes present a particular therapeutic challenge. Recent advances in percutaneous coronary intervention (PCI) may offer a new approach to treating this complex group of patients.
PCI started with balloon angioplasty, but this was associated with high RR. The introduction of ‘bare metal stents’ (BMS) improved clinical outcomes and lowered RR, but the treatment success of this procedure is still limited in high-risk populations. Drug-eluting stents (DES), which release antiproliferative agents, offer lower RR, and a reduced need for re-intervention. The most widely used DES to date are ‘Taxus’ (eluting paclitaxel) and ‘Cypher’ (eluting sirolimus).
The TAXUS V study - DES in complex cases
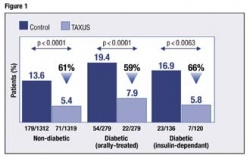
TAXUS V compared Taxus with a BMS specifically in higher risk groups, including patients with diabetes, small vessel disease, and where overlapping stents were required. The study included 356 diabetic patients. Over a 9-month follow up period, Taxus was found to have significantly lower RR (53% lower than BMS, P < 0.0001) and target lesion revascularisation (TLR) rates (46% lower than BMS, P < 0.09). In the sub-population of 102 insulin-dependent patients, RR was reduced by 55% and the need for TLR was reduced by 52%. This reduction was similar to that seen in the orally treated and non-diabetic population suggesting that Taxus has a similar significant benefit in orally-treated diabetic, insulin-dependent diabetic and non-diabetic populations.
Pooled analyses
A recent meta-analysis of four TAXUS clinical trials (TAXUS II, IV, V, and VI), involving 3,545 patients, including 814 diabetic patients, demonstrated clear superiority of Taxus over BMS for the overall population during 9-month follow up (in terms of TLR and RR). When diabetic and non-diabetic sub-populations were examined, TLR was similarly reduced in non-diabetic (61% reduction) and diabetic patients (59% and 66% for orally-treated and insulin-dependent patients).
Pooled data are also available for Cypher. This analysis included diabetic patients from the RAVEL, DIRECT, SIRIUS, E SIRIUS, and SVELTE studies. The TLR rate in all diabetic patients treated with Cypher was 5.8%, and the TLR rate for the insulin-dependent population was 10.1%.
While both DES offer significant benefits for diabetic patients, comparison of the Cypher and Taxus pooled analyses suggest that the Taxus stent may offer a small efficacy advantage in insulin-dependent diabetes.
Independent studies of DES in diabetes
A number of independent studies have also examined the use of DES in diabetic populations. These include ISAR-DIABETES, DIABETES, SIRTAX and PORTO. While these independent studies often provide valuable supporting data for the use of DES in patients with diabetes, their conclusions are sometimes limited by issues such as lack of statistical power, clinical primary endpoints, blinding, or randomization. Given the large amount of data in this fast moving field, recent guidelines for PCI from the European Society of Cardiology (ESC) recommend the use of DES based upon the level of available evidence from clinical trials. Both Cypher and Taxus are recommended for de-novo lesions in native coronary arteries. In addition, Taxus is recommended for long lesions. The ESC guidelines provide a useful tool for assessing the weight of evidence suggested by clinical trials in this field when selecting a DES.
Registries
Cardiology registries provide valuable insights into the meaning of clinical practices in the ‘real world’. ARRIVE, an FDA-mandated US registry of patients using Taxus, found that Taxus was associated with a 6-month re-intervention rate of 3.1% for diabetic patients, compared with 3.0% for non-diabetic patients. Similarly only small differences were found in the European MILESTONE II registry, where Taxus was associated with a re-intervention rate of 6.2% in diabetic patients compared with 5.5% for all patients at 12-month follow up. The E-Cypher registry demonstrated similar results in diabetic patients. Taxus and Cypher are the only two DES to have proven benefit in this high-risk group of patients in both randomized and large registry studies. These findings further support the application of DES in diabetic patients with CAD.
What next?
DES with improved deliverability are now being developed. These include Taxus Libertè, which is available in a range of diameters as low as 2.25 mm, launched in Europe in September 2005. These stents are designed to treat small, tortuous vessels that are otherwise very difficult to reach. This should further improve the treatment of CAD in diabetic patients who are known for their complex coronary anatomy. A paradigm shift away from coronary artery bypass grafts (CABG) to PCI as the first line approach for CAD in more complex patients will require convincing data from large, multicentre randomized controlled trials including SYNTAX, CARDia and FREEDOM. SYNTAX, which is ongoing and recruiting well, compares DES with CABG in approximately 4,200 patients with surgical-type disease in up to 90 centres worldwide. This study is expected to report its first data at the American Heart Association annual meeting in 2007.
Economics of using DES
When considering the financial case for PCI, it is vital to consider the follow up costs as well as the initial expense. Repeat interventions are very expensive to perform - in Europe CABG costs an average €13,000 and PCI costs approximately €3-5,000. High-risk patients are also at greater risk of needing re-intervention, increasing this cost still further.
Compared with BMS, the lower rate of re-intervention with DES means that costs are vastly reduced - in high-risk patients, the average 24-month follow up cost with Taxus was calculated as Ä625, compared with €1,960 for BMS. These cost savings associated with DES outweigh the higher initial procedural costs, with overall costs at 24 months for high-risk Taxus recipients calculated as €6,490, compared with Ä6,600 for BMS.
The financial case for using DES, rather than CABG, has recently been explored in two studies from Italy and Germany. In the Italian model, total health system costs were projected to decrease by 2.1% if 60% of PCI procedures were converted to DES, and 15% of CABG to DES. Studies such as SYNTAX will provide a stronger basis for economic analyses of DES vs CABG.
Conclusions
DES offer a major advance in the treatment of CAD in patients with diabetes and further improvements have already been made in DES with improved deliverability such as Taxus Liberté. Higher initial procedural costs for DES are expected to be compensated for by lower costs during follow-up, especially in patients at highest risk such as those with diabetes.
23.05.2006