Article • Controversial compound
Seeking alternatives to gadolinium-based contrast agents
Researchers are seeking alternatives to gadolinium-based contrast agents (GBCAs) to help raise levels of patient safety. An open hybrid session at ECR in Vienna heard how research across several centres has been examining the options of new approaches to reduce reliance on GBCAs.
By Mark Nicholls

Photo: supplied
In the session on “Safety in radiology: advancing medical imaging to the next level with EIBIR support”, Professor Rachel Katz-Brull discussed her project exploring the development of metal-free alternatives to GBCAs, examining the pros and cons of hyperpolarized MRI agents. In her presentation, she explained that there are 30 million doses of GBCAs used worldwide every year, with 8-11m examinations annually in the EU. While the chelate protects against the toxicity of the metal ion and provides specificity, there are remaining health risks with allergies or relatively rare adverse reactions.
Professor Katz-Brull, who heads the Center for Hyperpolarized MRI Molecular Imaging at the Hadassah Medical Center in Jerusalem, Israel, said: ‘There is gadolinium accumulation in bodies and brains of people, particularly of those who undergo multiple MRI examinations with contract-enhanced MRI. There are also consequences to the environment with rises in gadolinium levels in drinking water next to medical centres.’
Recommended article

Article • Energy conservation & waste reduction
Reducing the eco-footprint of radiology
Contrast agents in the wastewater and power-hungry imaging systems: The eco-footprint of healthcare is huge, and radiology departments are among the main culprits. An expert panel at the ECR Overture explored ways to make the field “greener”.
Pros and cons
As contrast enhanced MRI is a core element in identification and accurate diagnosis of tumours, inflammatory processes, and vascular conditions, the AlternativesToGd consortium targeted perfusion imaging and tissue-retention imaging in the search for alternatives to GBCAs. ‘There have been possible solutions by others, and these include use of other chelates or metal ions but we believe these are likely to encounter the same problems as gadolinium-based contrast agents,’ she said. ‘Our challenge is to find a mechanism for contrast-enhanced MRI that does not rely on metal ions.’
The consortium has targeted “hyperpolarized MRI agents” for this purpose. The pros, continued Professor Katz-Brull, are that they offer a high signal-to-background ratio and can be based on endogenous or inert compounds, yet the cons are that this is non-renewable magnetization and not given in trace amounts. She explained that the main challenges are the fast relaxation compared to the timeframes used in contrast-enhanced MRI, the generation of the hyperpolarized state, and the detection of hyperpolarized nuclei.
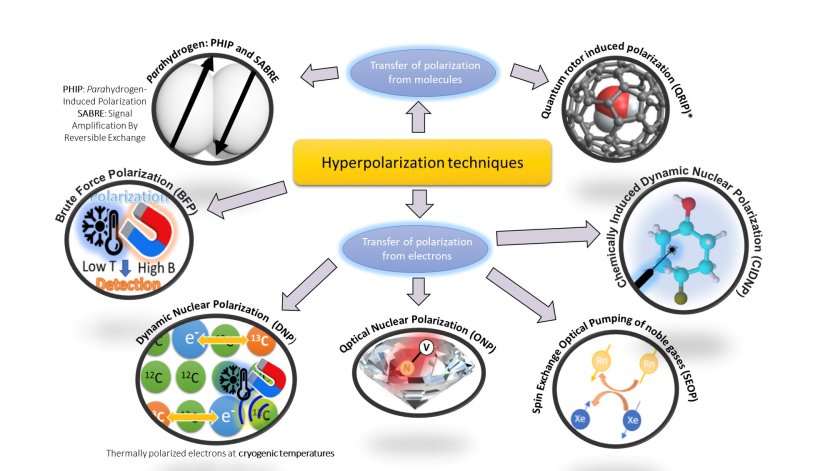
Image source: AlternativesToGd
Clinical settings
Professor Katz-Brull detailed the target of developing unique hyperpolarized agents for dDNP (dissolution Dynamic Nuclear Polarization) and administration routes that produce long lasting and intense signals in the blood pool. The work also focuses on the development of hyperpolarization that would be obtained using nano-diamonds and the development of tailored MRI tools for optimal observation of hyperpolarized agents in vivo in clinical settings. ‘Our ultimate goal,’ she said, ‘is to develop at least one replacement for the currently used GBCAs.’
She discussed the structure of the project covering technical and biological aspects, as well as utility and issues of safety, biodistribution, and tissue specificity.
Proof of concept
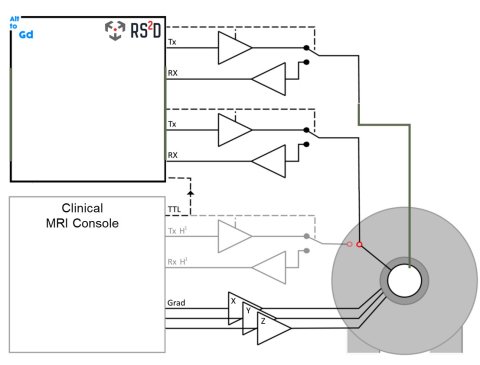
Image source: AlternativesToGd
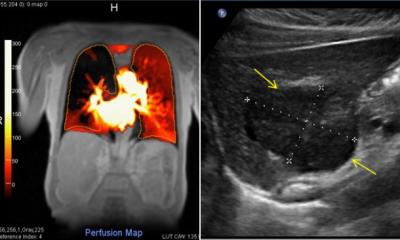
Within proof of concept, key areas included the first application of hyperpolarized water for angiographic applications in large animals; perfusion imaging with hyperpolarized 15N-labelled urea with dramatically improved sensitivity; demonstration of perfusion tissue retention using hyperpolarized nitrate as a contrast agent; demonstration of a prolonged visibility window for hyperpolarized water; and visualisation of perfusion MRI, and of small molecules hyperpolarized by PHIP-SAH (ParaHydrogen Induced Polarization-Side Arm Hydrogenation) and via polarization transfer from hyperpolarized 13C containing nano-diamonds. Creation of these new contrast agents for MRI has involved combining multiple and diverse expertise to look at new imaging probes for perfusion, nano-capsules for hyperpolarized materials, efficient sampling strategies, detection of hyperpolarized 15N urea, validation, and designing new MRI tools for hyperpolarized x-nuclei.
Medical reality
Explaining challenges and success, she said that with nano-diamonds, the project has found ‘many interesting and new insights’ about the surface of nano-diamonds and the importance for the polarization process. ‘We hope that this will bring this technology closer to medical reality,’ she added. Her research partners, she continued, have been successful in measuring 13C urea on a clinical scanner using a vendor independent transmit/receive system that they have built for the project.
Project achievements so far to replace GBCAs include the establishment of a pre-clinical model of stroke for future assessment of perfusion alterations using hyperpolarized probes, a new analysis model that combines assessment of metabolism and perfusion, sufficient polarization achieved by the PHIP-SAH method to acquire in vitro and in vivo images of 13C, and establishing that Seiffert-Spirals trajectories are useful for hyperpolarized 13C imaging in a clinical scanner.
Supporting science
The session was co-organised with EIBIR (European Institute for Biomedical Imaging Research), a not-for-profit liability company founded by ESR supporting scientist performing research, particularly in areas of safety improvement. It also heard from Alexandru Dasu from the Skandion Clinic in Uppsala, Sweden, on the SINFONIA project, looking at radiation risk appraisal for detrimental effects from radiation exposure during the management of patients with cancer and from EIBIR project officer Peter Gordebeke on EIBIR activities and funding opportunities.
Profile:
Professor Rachel Katz-Brull heads the Center for Hyperpolarized MRI Molecular Imaging at the Hadassah Medical Center in Jerusalem where a major focus is on developing new diagnostics and improve existing diagnoses using hyperpolarized molecular imaging agents in MRI. She is an Associate Professor of Imaging at the Hebrew University Faculty of Medicine and has more than 28 years of experience in the field of biomedical nuclear magnetic resonance imaging and spectroscopy.
21.07.2022