Article • An estimated 800,000 deaths in 2030
Warning of looming global liver disease pandemic
Based on recent studies of liver diseases, Professor Dina Tiniakos, pathologist at the National and Kapodistrian University of Athens, concludes that cases will soar worldwide by 2030, with 800,000 liver deaths related to NASH (Non-alcohol related steatohepatitis), costing health economies multi-billions of dollars.
During her presentation ‘From the origins of NASH to the future of metabolic fatty liver disease’, she outlined concerns in the keynote Symeonidis Lecture to the 33rd European Congress of Pathology. Tiniakos explained how NAFLD (Non-alcoholic fatty liver disease) – which includes NASH – is a complex metabolic liver disease with increasing prevalence.
Common causes

Abdominal obesity and type 2 diabetes are among common causes of a condition, which is now also referred to under a new definition of MAFLD (metabolic, dysfunction-associated fatty liver disease) to also include evidence of hepatic steatosis. In lean patients, that includes at least two metabolic risk abnormalities, such as prediabetes, insulin resistance, hypertension and increased triglycerides.
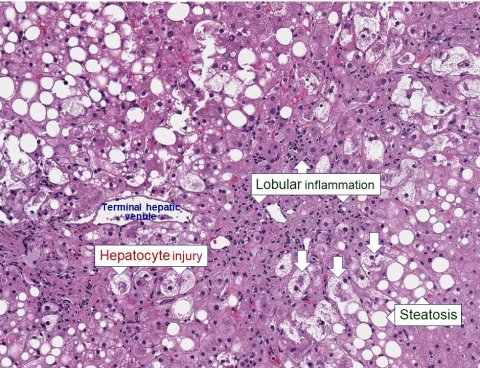
From simple NAFLD, some patients will progress to steatohepatitis (NASH) with liver inflammation and hepatocyte injury, of which 40% may show progressive fibrosis, and 3-5% of NAFLD patients may progress to cirrhosis.
Detailing the history of the condition, Tiniakos said NAFLD has been overlooked for a long time – the term NASH did not exist in medical vocabulary until the 1980s. ‘Because NAFLD has been overlooked for decades may be the reason that currently there is a lack of NASH-specific drugs and reliable biomarkers,’ she added.
Liver biopsy
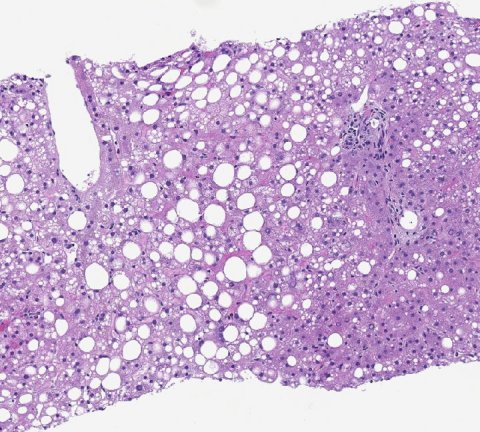
Fatty liver disease was first recognised in the 19th century, and became better identified due to liver biopsy used from the late ’50s. Further recognition and understanding of NAFLD pathogenesis developed in the last three decades. ‘Today,’ Tiniakos continued, ‘liver biopsy is still the most accurate method for diagnosing NASH and classifying NAFLD, but the natural history of NAFLD is dynamic and the disease may wax and wane, depending on diet, exercise and other factors. As disease progresses, fibrosis develops, and 20% of patients may be rapid progressors and may develop advanced fibrosis faster. It is important to identify rapid progressors to ensure they have the right management.’
Non-invasive diagnosis
There are, however, steps towards non-invasive diagnosis of NASH and fibrosis in NAFLD, including serum biomarkers, imaging methods to assess liver fibrosis and sequential combination of imaging with scores. In primary care, non-invasive methods are useful, Tiniakos pointed out, to select patients at low risk of advanced fibrosis and while they can predict long-term outcomes and reduce need for liver biopsy – from 33 to 19 per cent – they cannot substitute the need for biopsy. The professor warned that the future of metabolic liver disease looks worrying and global health systems should prepare for a NASH epidemic. This follows a ‘call for action’ by global experts to ‘prepare for the NASH epidemic’.
NASH prevalence is in 1.5-6.5 percent of the population, but this is expected to increase to 63% by 2030. Up to 21 million Americans and 100 million individuals worldwide have NASH.
Future developments
NASH-related advanced fibrosis patients are likely to double by 2030; there will be 800,000 NASH-related liver deaths by 2030 and $95.4 billion will be the direct cost of NASH in the USA in the next two decades. Whilst there are currently no approved drugs for treating NASH, more than 200 clinical trials are in progress.
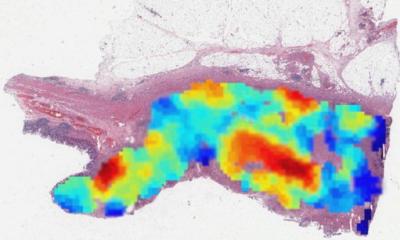
Tiniakos also believes that pathological grading and staging of NAFLD/NASH in clinical trial settings in the near future may be supported by artificial intelligence methods. She also pointed to advances in microscopy techniques such as second-generation harmonic generation/two photon excitation fluorescence imaging (SHG/TPE), and stain-free microscopy methods may improve reproducibility and standardisation of liver biopsy assessment.
In indicating that the future focus must be on establishing preventive methods, discovering accurate non-invasive diagnostic and prognostic biomarkers, and developing effective treatment for individuals with advanced NASH, Tiniakos said liver pathologists will continue to play a central role in this effort.
Profile:
Professor Dina Tiniakos works in the Department of Pathology at Aretaieion Hospital, the National and Kapodistrian University of Athens, Greece, and the Translational and Clinical Research Institute in the Faculty of Medical Sciences at Newcastle University, UK. She is also Past President of the European Society of Pathology.
17.11.2021