Source: Mayo Clinic
News • In children
Treating scoliosis with vertebral body tethering
Fusion surgery has been the long-standing treatment for people with scoliosis - a side-to-side curve of the spine. But other options have become available — including vertebral body tethering for children with scoliosis.
"Vertebral tethering is a new tool in the tool kit for the treatment of scoliosis," says A. Noelle Larson, M.D., a pediatric orthopedic surgeon at Mayo Clinic in Rochester, Minnesota, and expert in scoliosis surgery.
Its cause is unknown, but scoliosis affects an estimated 1 in 300 people worldwide. A spinal curve greater than 10 degrees is defined as scoliosis. Severe scoliosis that requires treatment is more common in women. Mild scoliosis cases are monitored over time. Small curves in growing children can progress rapidly without treatment. Moderate scoliosis in growing children is treated with bracing. Until recently, severe scoliosis could only be treated with fusion surgery.
With fusion surgery, surgeons connect the bones in the spine (vertebrae) through an incision in the back and place screws in the spine. Rods are attached to the screws to pull the spine into a corrected position, and then those vertebrae are fused into a solid sheet of bone.
"Fusion had been a reliable treatment approach with a durable long-term result and powerful correction of the spinal curvature," Dr. Larson says. "But with fusion, the spine no longer grows, and there is no flexibility of the spine over the fused vertebrae. Some patients and families value spinal motion and growth, and would prefer another treatment approach for severe scoliosis."
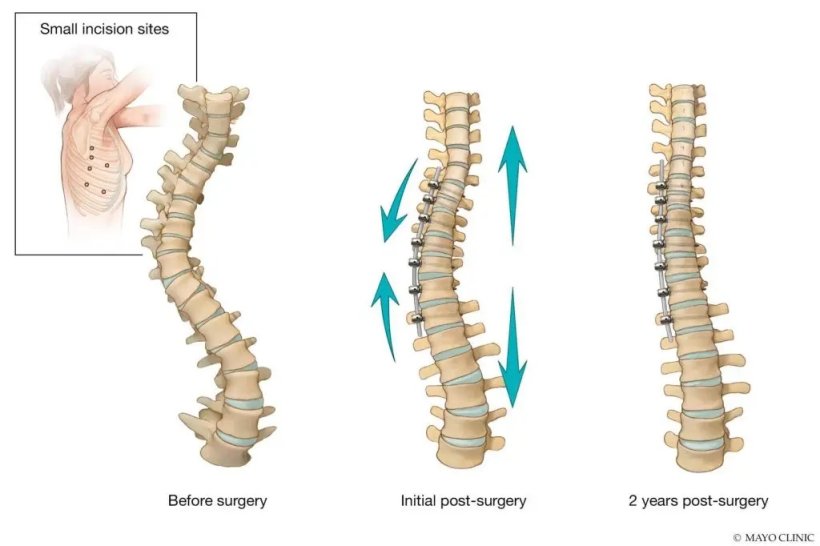
In August 2019, the Food and Drug Administration (FDA) granted humanitarian device exemptions for two nonfusion surgical implants — vertebral body tethering and posterior dynamic distraction surgery. This is a more limited approval which cites the safety of the procedure, but not the effectiveness of it. A flexible cord is placed in the front of the spine through small incisions and anchored to the vertebrae with screws placed through 1-inch incisions.
When the cord is tightened during the surgery, the spine straightens. As the child grows, the spine may straighten even more. Growing children with moderate to severe scoliosis and certain curve types are eligible for the procedure.
Although spinal motion and growth remains, there are potential downsides to the procedure. At this time, there is a higher risk for a second surgery compared with fusion surgery, due to overcorrection or undercorrection of the scoliosis. The long-term durability of the procedure is not known, and a fusion surgery may be required eventually.
Since the FDA approval, more studies on vertebral tethering are underway nationally. Dr. Larson and her colleague Todd Milbrandt, M.D., are leading an FDA-approved study to assess the results of vertebral body tethering at two years after surgery. Dr. Larson also is the Mayo principal investigator for international scoliosis registries from the Setting Scoliosis Foundation and Pediatric Spine Foundation for patients treated with vertebral body tethering.
"Patients and families should be informed about their treatment options," Dr. Larson says. "Some families would like an alternative to fusion surgery. More information is coming out about nonfusion scoliosis surgery. If patients are interested in vertebral body tethering, I would encourage them to go to an experienced center, as this is an emerging technology."
Source: Mayo Clinic
24.02.2022