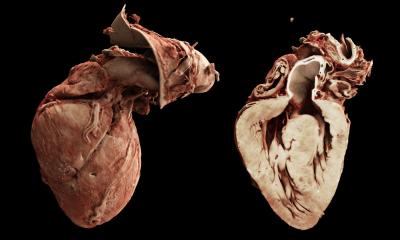
Article • Computer & computed tomography
The virtual-heart arrhythmia risk predictor
Research by a team at John Hopkins University (JHU) in Baltimore, USA highlights the patients who are most likely to face lethal arrhythmias. They have developed a personalised 3-D virtual heart that can help predict the risk of sudden cardiac death.
Report: Mark Nicholls

When patients suffer arrhythmia, cardiologists often respond by fitting a small defibrillator implant to sense the onset of arrhythmia and jolt the heart back to a normal rhythm. However, the question remains over how they decide which patients need the implant and the invasive surgery needed to fit it.
Aiming to address this, a team from the Department of Biomedical Engineering and Institute for Computational Medicine at JHU has developed a non-invasive 3-D virtual heart assessment tool to help doctors determine which patients face the highest risk of a life-threatening arrhythmia and would benefit most from a defibrillator implant.
Early evidence suggests the new digital approach yielded more accurate predictions than the current blood pumping measurement used by most physicians. ‘Our virtual heart test significantly outperformed several existing clinical metrics in predicting future arrhythmic events,’ Professor Natalia Trayanova, the university’s inaugural Murray B. Sachs Professor of Biomedical Engineering, said. ‘This non-invasive and personalised virtual heart-risk assessment could help prevent sudden cardiac deaths and allow patients who are not at risk to avoid unnecessary defibrillator implantations.’
A pioneer in developing personalised imaging-based computer models of the heart, she has worked on the project with cardiologist Katherine C Wu, associate professor in the Johns Hopkins School of Medicine, whose research has focused on MR resonance imaging approaches to improving cardiovascular risk prediction.
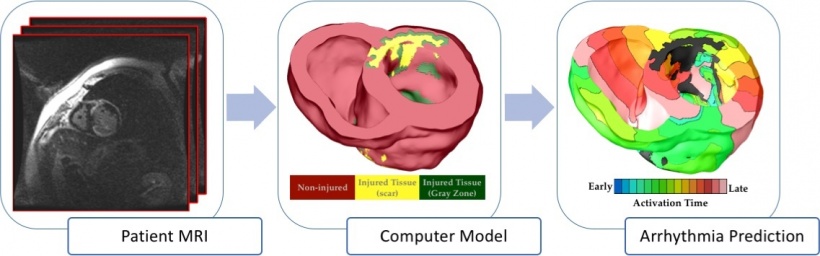
For the study, Trayanova’s team formed its predictions by using the distinctive magnetic resonance imaging (MRI) records of patients who had survived a heart attack but were left with damaged cardiac tissue that predisposes the heart to deadly arrhythmias. The study involved data from 41 patients who had survived a heart attack and had an ejection fraction – a measure of how much blood is being pumped out of the heart – of less than thirty-five percent.
Patients in this range are usually recommended implantable defibrillators, however, with the JHU team concerned about this measuring score system, they invented an alternative to these scores by using pre-implant MRI scans of the recipients’ hearts to build patient-specific digital replicas of the organs.
Using computer-modelling techniques developed in Trayanova’s lab, the geometrical replica of each patient’s heart was brought to life by incorporating representations of the electrical processes in the cardiac cells and the communication among cells. In some cases, the virtual heart developed an arrhythmia, and in others it did not.
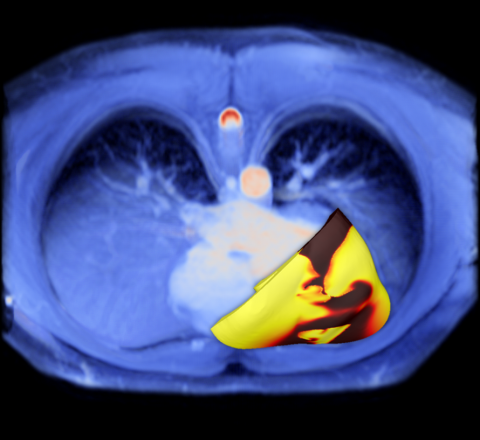
The new non-invasive way to gauge the risk of sudden cardiac death due to arrhythmia has been named the VARP (virtual-heart arrhythmia risk predictor). Subsequent tests showed that patients who tested positive for arrhythmia risk by VARP were four times more likely to develop arrhythmia than those who tested negative. VARP predicted arrhythmia occurrence in patients four-to-five times better than the ejection fraction and other, invasive and non-invasive, existing clinical risk predictors. ‘We demonstrated that VARP is better than any other arrhythmia prediction method out there,’ Trayanova confirmed. ‘By accurately predicting which patients are at risk of sudden cardiac death, the VARP approach will provide the doctors with a tool to identify those patients who truly need the costly implantable device, and those for whom the device would not provide any life-saving benefits.’
Wu said that the early results indicate the more nuanced VARP technique could be a useful alternative to the one-size-fits-all ejection fraction score.
Trayanova is hopeful the new risk prediction methodology could also be applied to patients who had prior heart damage, but whose ejection fraction score did not target them for therapy under current clinical recommendations. The next step is to conduct further tests involving larger groups of heart patients.
Profile:
Natalia Trayanova PhD is the John Hopkins University’s inaugural Murray B. Sachs Professor of Biomedical Engineering in the Department of Biomedical Engineering and Institute for Computational Medicine. She received her doctorate at the Bulgarian Academy of Sciences in Sofia (1986) and post-doctoral training in biomedical engineering at Duke University. Her research focuses on understanding the normal and pathological electrophysiological and electromechanical behaviour of the heart.
25.08.2016